Name the following alkenes, and predict the products of their reaction with (1) meta-chloroperoxybenzoic acid, (ii) KMnO4
Question:
Name the following alkenes, and predict the products of their reaction with (1) meta-chloroperoxybenzoic acid, (ii) KMnO4 in aqueous acid, and (iii) O3, followed by Zn in aceticacid:
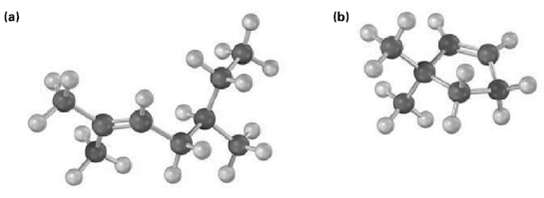
Transcribed Image Text:
(a) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a CH3 CH3CCHCHCHCHCH3 25Dimethyl2heptene CH3 RCO3H KM...View the full answer

Answered By

Vincent Omondi
I am an extremely self-motivated person who firmly believes in his abilities. With high sensitivity to task and operating parameters, deadlines and keen on instructions, I deliver the best quality work for my clients. I handle tasks ranging from assignments to projects.
4.90+
109+ Reviews
314+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Name the following alkynes, and predict the products of their reaction with (i) H2 in the presence of a Lindlar catalyst and (ii) H3O+ in the presence ofHgSO4: (b) (a)
-
Predict the products formed by periodic acid cleavage of the following diols. (a) CH3CH(OH)CH(OH)CH3 (b) (c) (d) CHAOH OH OH Ph-CCH OH)CH,CH CH HO HO
-
Name each of the following alkenes or alkynes. a. CH2 = CH-CH2-CH3 b. c. d. e. f. g. CH3 C-CH-CH3 CH3 CH CH3 CH3CH2CH CH CH CH CH3 CH, C-CH-CH CH, CH2-CH, CH3 CH2CHs CH, CH2CH3 CH3 C C-CH CH3 CH3
-
Consider a process consisting of five resources that are operated eight hours per day. The process works on three different products, A, B, and C; Resource Number of Workers Processing Time for A...
-
Sarpedon Corp. claims that its car batteries average at least 880 CCA (cold-cranking amps). Tests on a sample of 9 batteries yield a mean of 871 CCA with a standard deviation of 15.6 CCA. (a) State...
-
Consider the linear discrete-time dynamical system yt+1 = 1. 0 + m(yt - 1.0). For each of the following values of m, a. Find the equilibrium. b. Graph and cobweb. c. Compare your results with the...
-
What are the benefits of using value management in projects?
-
The dollar-value LIFO method was adopted by Enya Corp. on January 1, 2014. Its inventory on that date was $160,000. On December 31, 2014, the inventory at prices existing on that date amounted to...
-
Consider the following pseudocode for calculating ab, where a and b are positive integers. 19 FastPower Input: positive integers a and b. Output: ab. return a if b=1 then else c:=b.b...
-
MGM International operates casinos and resorts across the U.S. and in China. The company reported the following in its SEC filings. We maintain an allowance for doubtful casino accounts at all of our...
-
One of the chain-termination steps that sometimes occurs to interrupt polymerization is the following reaction between two radicals. Propose a mechanism for the reaction, using fishhook arrows to...
-
Draw the structures of alkenes that would yield the following alcohols on hydration (red = O). Tell in each case whether you would use hydroboration/oxidation oroxymercuration. (b) (a)
-
Determine whether each of the following pairs of events is independent: a. Rolling a pair of dice and observing a 2 on one of the dice and having a total of 10 b. Drawing one card from a regular deck...
-
Jenna is a single taxpayer. During 2 0 1 8 , she earned wages of $ 1 3 1 , 0 0 0 . She does not itemize deductions, so she will take the standard deduction to calculate 2 0 1 8 taxable income. In...
-
What is the value today of a money machine that will pay $ 2 , 5 5 2 . 0 0 every six months for 1 0 . 0 0 years? Assume the first payment is made six months from today and the interest rate is 1 0 ....
-
Assume the following excerpts from a company s balance sheet: Beginning Balance Ending Balance Bonds payable $ 5 0 0 , 0 0 0 $ 6 5 0 , 0 0 0 Common stock $ 9 5 0 , 0 0 0 $ 9 5 0 , 0 0 0 Retained...
-
Rose Hill, a soybean farm in northern Minnesota, has a herd of 3 3 dairy cows. The cows produce approximately 1 , 8 2 0 gallons of milk per week. The farm currently sells all its milk to a nearby...
-
Contribution Income Statement, Cost - Volume - Profit Graph, and Taxes Jail and Sail: Alcatraz Tour and Cruise provides sunset sightseeing tours of Alcatraz and the San Francisco Bay. Tickets cost $...
-
Where is the oldest rocknear a spreading center or far away from it? Explain your thinking.
-
Prove the result that the R 2 associated with a restricted least squares estimator is never larger than that associated with the unrestricted least squares estimator. Conclude that imposing...
-
What happens to the vapor pressure of a substance when its surface area is increased at constant temperature? (a) The vapor pressure increases. (b) The vapor pressure remains the same. (c) The vapor...
-
A student, Flick Flaskflinger, in his twelfth year of graduate work, needed to prepare ethylmagnesium brdmide from ethyl bromide and magnesium, but found that his laboratory was out of diethyl ether....
-
When sec-butylbenzene undergoes free-radical bromi-nation, one major product is formed, If the starting material is optically active, predict whether the substitution product should also be optically...
-
Three alkyl halides, each with the formula C7HBr, have different boiling points. One of the compounds is optically active. Following reaction with Mg in ether, then with water, each compound gives...
-
Watch the video "IDEO shopping cart project https://www.youtube.com/watch?time_continue=5&v=M66ZU2PCIcM what are the attributes in the IDEO organization that encourage and enhance innovation in an...
-
1 Critically evaluate the importance of the media in influencing partners, opinion formers, stakeholders, customers and the public at large. 2 Critically assess domestic, national and international...
-
A 0.7 MeV photon scatters from an electron initially at rest. If the photon scatters at an angle of 35, calculate (a) the energy and wavelength of the scattered photon, (b) the kinetic energy of the...

Study smarter with the SolutionInn App


