Name the following alkenes, including the cis or Tran?s designation: (b)
Question:
Name the following alkenes, including the cis or Tran?s designation:
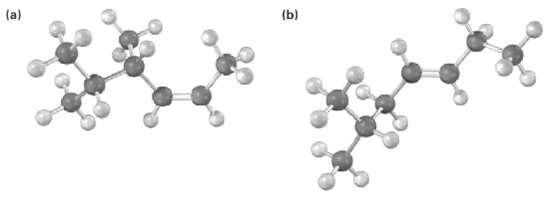
Transcribed Image Text:
(b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
H3 CH3 CH3CHCH CH...View the full answer

Answered By

Leah Muchiri
I am graduate in Bachelor of Actuarial Science and a certified accountant. I am also a prolific writer with six years experience in academic writing. My working principle are being timely and delivering 100% plagiarized free work. I usually present a precised solution to every work am assigned to do. Most of my student earn A++ GRADE using my precised and correct solutions.
4.90+
52+ Reviews
125+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Name each of the following alkenes or alkynes. a. CH2 = CH-CH2-CH3 b. c. d. e. f. g. CH3 C-CH-CH3 CH3 CH CH3 CH3CH2CH CH CH CH CH3 CH, C-CH-CH CH, CH2-CH, CH3 CH2CHs CH, CH2CH3 CH3 C C-CH CH3 CH3
-
Name each of the compounds below. Use cis / trans and/or E/Z designations, if appropriate, to designate stereochemistry a. b. c. Cl C=C H,C CH3 CH2CI H3C H3C H,C Cl
-
The name Computer Emergency Response Team is the historic designation for the first team (CERT/CC) at Carnegie Mellon University (CMU). CERT is now a registered service mark of Carnegie Mellon...
-
Suppose the money supply is $250 million dollars and the demand for money is given by Qm D = 400 - 40i, where Qm D is in millions of dollars. A. What is the equilibrium interest rate in this economy?...
-
Which of the four path-goal styles do you think would be the best for managing a group of software engineers? Justify your answer.
-
Deanna is 62 years old. She plans to retire in 3 years. She has $300,000 in a savings account that yields 2.25% interest com- pounded daily. She has calculated that her final working year's salary...
-
What is Engineering?
-
Brian Cleary and Rita Burke filed a suit against the major cigarette maker Philip Morris USA, Inc., seeking class-action status for a claim of deceptive advertising. Cleary and Burke claimed that...
-
At what temperature would one mole of molecular oxygen (o2) have 5.0 x 10 3 J of translational kinetic energy? Note: The atomic mass of O is 16 u. Show work.
-
Frank and Dale Cumack are married and filing a joint 2017 income tax return. During 2017, Frank, age 63, was retired from government service and Dale, 55, was employed as a university instructor. In...
-
Which of the following compounds can exist as pairs of cisTrans isomers? Draw each cisTrans pair, and indicate the geometry of each isomer. (a) CH3CH = CH2 (b) (CH3)2C = CHCH3 (c) CH3CH2CH = CHCH3...
-
Which member in each of the following sets has higher priority? (a) H or Br (b) C1 or Br (c) CH 3 or CH 2 CH 3 (d) NH 2 or OH (e) CH 2 OH or CH 3 (f) CH 2 OH or CH = O
-
What is an internal control framework, and how is it used in a financial statement audit?
-
Record adjusting entries for the following information. Merchandise inventory Store supplies 33,600 1,915 975 Office supplies Prepaid insurance 5,255 Equipment 63,490 Accumulated depreciation,...
-
Bunnell Corporation is a manufacturer that uses job-order costing. On January 1, the company's inventory balances were as follows: Raw materials Work in process Finished goods $ 59,500 $ 37,600...
-
On January 1 of Year 1, Innovative Lab issued a 4-year $50,000 note to a local bank with fixed interest payments based on 6%, payable annually on December 31. To hedge the risk of a fixed interest...
-
If I invest a single amount of $14,000 in an account earning 8% p.a. compounding quarterly for 5 years, how much interest will I have earned in those 5 years?
-
Manvir had to make payments of $1,125 every 6 months to settle a $22,000 loan that he received at 4.52% compounded semi-annually. a. How long did it take to settle the loan?
-
Do surface currents affect water temperature, air temperature, or both?
-
When a company has a contract involving multiple performance obligations, how must the company recognize revenue?
-
Nitrogen has a normal boiling point of 77.3 K and a melting point (at 1 atm) of 63.1 K. Its critical temperature is 126.2 K, and its critical pressure is 2.55 * 10 4 torr. It has a triple point at...
-
Draw the structure of 4-isopropyl-2,4,5-trimethylheptane.
-
What is the dissociation constant of an acid that has a pKa of (a) 4 (b) 7.8 (c) -2
-
Using the pka values in Table 3.1, calculate the equilibrium constant for the following reaction. F- acting as a base toward the acid HCN
-
Consider a 15%, 20 year bond that pays interest annually, and its current price is $850. What is the promised yield to maturity?
-
You are a dual-income, no-kids family. You and your spouse havethe following debts (total): mortgage, $212,000; auto loan,$22,000; credit card balance, $16,000; other debts, $22,000.Further, you...
-
Can you please provide a feedback to this post? 1. What is the managerial view on business? What are the stakeholders view on business? How do they differ? The management viewpoint on business...

Study smarter with the SolutionInn App


