Name the following ethers: (a) (b) CH CH OCH2CH2CH3 CHH H (c) (d) Br C (e)
Question:
Name the following ethers:
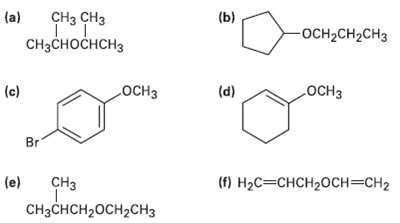
Transcribed Image Text:
(a) (b) CHз CHз OCH2CH2CH3 CHзсноснсHз ОСHЗ ОСНЗ (c) (d) Br CНз (e) () Нас—снCH20сн%3сH2 CH3CHCH20CH2CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (18 reviews)
Ethers can be named either as alkoxysubstituted compounds or by citing the two group...View the full answer

Answered By

Talha Talib
I am a member of IEEE society. As i am a student of electrical engineering badge 17 but beside of this i am also a tutor in unique academy. I teach calculus, communication skills, mechanics and economics. I am also a home tutor. My student Muhammad Salman Alvi is a brilliant A-level student and he performs very well in academics when i start to teach him. His weak point was mathematics but now he is performing well in mathematics. I am a scholarship holder in Fsc as i scored 1017 marks in metric out of 1100. Later on i got scholarship in Punjab Group of Colleges. I got 2nd position in robotics competition in 2018 as my project home automation select for the exhibition in Expocentre.
4.60+
23+ Reviews
62+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
One way of naming ethers is to name the two alkyl groups attached to the oxygen atom in alphabetical order and add the word ether. If the two alkyl groups are the same, we use the prefix di-, for...
-
Ethers tend to dissolve in alcohols and vice versa. Represent the hydrogen-bonding interaction between an alcohol molecule and an ether molecule.
-
Ethers are not easily differentiated by their infrared spectra, but they tend to form predictable fragments in the mass spectrum. The following compounds give similar but distinctive mass spectra....
-
What is the result of the following? A. 3.011459 B. 3.1401MM C. 59.011459 D. 59.1401MM E. The code does not compile. F. The code compiles but throws an exception at runtime. import java.time. *;...
-
Oliver is a U.S. citizen employed by a multinational corporation at its London office. Oliver is married to Regina, a British citizen, and they reside in England. Regina receives substantial rent...
-
Data since 2000 reveals a moderately strong positive correlation between the federal debt and attendance at National Football League games. Would restricting the number of persons attending games...
-
What is the hedonic wage function? What does the slope of the hedonic wage function measure?
-
The Vernom Corporation produces and sells to wholesalers a highly successful line of summer lotion and insect repellents. Vernom has decided to diversify to stabilize sales throughout the year. A...
-
How much work ( in joules) is done in lifting a 45.57 newton box from the floor to a table that is 0.73 meters above the floor? Round your final answer to two decimal places. A ball is thrown...
-
Explain the role of secondary data in gaining customer insights. Where do marketers obtain secondary data, and what are the potential problems in using such data?
-
Would you expect p-methyl phenol to be more acidic or less acidic than un-substituted phenol? Explain.
-
Why do you suppose only symmetrical ethers are prepared by the sulfuric acid-catalyzed dehydration procedure? What product(s) would you expect if ethanol and 1-propanol were allowed to react...
-
What is type inferencing, as used in ML?
-
What is the end value of investing $27,500 for 7 years at an annual rate of 14%. Interest is compounded semi-annually.
-
Mr. Knight still can't find some of his enthalpy values. The last equation on his list is this: CHC130) + 3HCl(g) CH4(g) + 3Cl2(g) Calculate the enthalpy value for this reaction using the following...
-
Problem 4 (20 points): A consulting firm, Omega Consulting, is considering offering a data analysis service. If the demand is high (i.e., there is a favorable market for the service), the firm could...
-
You will work with the production function: FpK, Lq" AKL 1. Let K be capital, L labor, w is the wage (the price of one unit of labor), and r is the rental rate (the price of one unit of capital. What...
-
Starting with an investment in 2009 , what would have been the return over the subsequent 5 years? Assume you reinvest all dividends at the end of each year. What is the annualized HPR for the 5-year...
-
Based on Aors notes from her previous meeting with Njau, the behavioral consideration exhibited by Njau is most likely: A. a consumption gap. B. the annuity puzzle. C. heightened loss aversion. Nomie...
-
What are three disadvantages of using the direct write-off method?
-
Which has the greater potential to produce more available work steam at 2 MPa and 800 C or steam at 1.4 MPa and 900 C?
-
Explain which of these bonds has the absorption for its stretching vibration at higher wave number: (a) C H or C D (b) C = C or C C (c) C C1 or C I
-
Indicate the positions of the absorption bands and any other noteworthy features in the hydrogen region of the IR spectra of thesecompounds: CH3 ) C,CH b) NH2 d) CH2=CHCH,CH,OH f) CH,CH,CH,CH e)...
-
Explain why the presence of a triple bond is much easier to detect in the IR spectrum of 1-hexyne than it is in the spectrum of3-hexyne. 80 60 O The sp-hybridized CH absorption bands: from 3000-2850...
-
www The sugar cane industry is a big industry on Hawaii. Raw sugar cane is first cut from the fields, then chopped and shredded. The raw cane contains 15 wt% sucrose, 25 wt% solids, and water, along...
-
What does a manager who subscribes to Theory X believes ? Explain thoroughly.
-
Keri, in forming a new corporation, transfers land to the corporation in exchange for 100 percent of the stock of the corporation. Keri's basis in the land is $295,000, and the corporation assumes a...

Study smarter with the SolutionInn App


