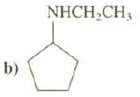
Name these compounds: (a) CH 3 CH 2 CH 2 NH 2 b) NHCH,CH,
Question:
Name these compounds:
(a) CH3CH2CH2NH2
Transcribed Image Text:
b) NHCH,CH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
In the common names of amines the suffix amine is appended to the name of t...View the full answer

Answered By

Madhvendra Pandey
Hi! I am Madhvendra, and I am your new friend ready to help you in the field of business, accounting, and finance. I am a College graduate in B.Com, and currently pursuing a Chartered Accountancy course (i.e equivalent to CPA in the USA). I have around 3 years of experience in the field of Financial Accounts, finance and, business studies, thereby looking forward to sharing those experiences in such a way that finds suitable solutions to your query.
Thus, please feel free to contact me regarding the same.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Name each of the following compounds using R,S and E,Z (Section 3.5) designations where necessary: a. b. c. d. e. f. g. h. CH2CH3 H- CH3 H2CH C-C C-C CI Br CH,CH CH-CH,CH,CH, - H3C CH2CH2l CH2CH...
-
Name each of the following compounds according to substitutive IUPAC nomenclature: (a) (CH3)2CHCH2CH2CH2Br (b) (CH3)2CHCH2CH2CH2OH (c) Cl3CCH2Br (d) (e) CF3CH2OH (f) (g) (h) (i) Cl2CHCHBr CI OH ,
-
Name each of the compounds below. Use cis / trans and/or E/Z designations, if appropriate, to designate stereochemistry a. b. c. Cl C=C H,C CH3 CH2CI H3C H3C H,C Cl
-
Explain factors governing recruitment?
-
Silver water Ltd manufactures sophisticated six-tuner personal video recorders and has decided to develop an activity-based product costing system to assign labour and overhead costs to products. The...
-
Is the current privacy protection for e-mail and internet use sufficient?
-
What is the System Element Architecture (SEA) Model?
-
Sam Hutchins is planning to operate a specialty bagel sandwich kiosk but is undecided about whether to locate in the downtown shopping plaza or in a suburban shopping mall. Based on the following...
-
Scalia Systems manufactures rugged handheld computers for use in adverse working environments. Scalia tries to maintain inventory at 40% of the following month's expected unit sales. Scalia began the...
-
The Future Government Club wants to sponsor a panel discussion on the upcoming national election. The club wants four of its members to lead the panel discussion. Obtain a simple random sample of...
-
Name thesecompounds: a) CHOCHCH3 OCH, b) CI
-
Name these compounds: CH3 a) CHCHCHCHCHCH, b) NHCH, NH
-
The following situations are independent of each other. Instructions For each situation, prepare the appropriate journal entry for the redemption of the bonds. a. Mikhail Corporation redeemed \(\$...
-
Consider the following function. F(t) = 3t5 - 5t + 22 Find the derivative of the function. F'(t) = 15/ - 15/4 Find the critical numbers of the function. (Enter your answers as a comma-separated...
-
Consider the relation R = {(a, b) ENXN| b = 2a +1}. Which pairs belong to R? a. (1,3) b. C. (2,8) d. (1,7) e. (1,5) f. (10, 21) 8. (2,5) (3,8) (3,1) j. (3,7) Of. h. (10, 11) i.
-
1.a) For the original tree given below. write down the preorder, inorder and postorder traversals. 44 26 35 14 33 31 10 42 19 27 b) With the help of examples, explain what is the lower bound and what...
-
Find d'y dx if -2x + 7y4 10.
-
Consider the following graph a. Find all the simple paths from node A to node E b. Find indeg(D) and outdeg(D) c. Are there any sources or sinks d. Find the adjacency matrix for the graph
-
Describe life-cycle budgeting and life-cycle costing and when should companies use these techniques?
-
A supermarket chain is interested in exploring the relationship between the sales of its store-brand canned vegetables (y), the amount spent on promotion of the vegetables in local newspapers (x1)...
-
Find an equation of the line through the points (1, 2) and (3, 4).
-
Trans-2-Buterw is more stable than cic-2-hutene by only 4kJ/mol, but trans-2, 2, 5; 5-tetramethyl-3-hexene is more stable than its cis isomer by 3kJ/mol. Explain.
-
Cyclodecene can exist in both cis and Trans forms, but cyclohexene cannot. Explain. (Making molecular models is helpful.)
-
Normally, a Trans alkene is more stable than its cis isomer Trans-Cyclooctene, however, is less stable than cis-Cyclooctene by 38.5kJ/mol. Explain.
-
Question: Read the document on this link: https://pui2022q4.tiiny.site/ 1. You form part of a company in South Africa and you have to continuously do business locally and internationally on the...
-
Banjo Education Corp. issued a 4%, $80,000 bond that pays interest semiannually each June 30 and December 31. The date of issuance was January 1, 2020. The bonds mature after four years. The market...
-
A projectile if fired from 19.6m above the ground with an initial speed of 55.6m/s at an angle of 41.2 degrees above the horizontal. a) Determine the time necessary for the projectile to reach its...

Study smarter with the SolutionInn App


