Nitrites (RC ? N) react with Grignard reagents (R'MgBr). The reaction product from 2-methyipropanenitrile with methyl magnesium
Question:
Nitrites (RC ? N) react with Grignard reagents (R'MgBr). The reaction product from 2-methyipropanenitrile with methyl magnesium bromide has the following spectroscopic properties. Propose a structure.
MS: M+ = 86
IR: 1715 cm?1?
1H NMR: 1.05 ? (6 H, doublet, J = 7 Hz); 2.12 ? (3 H, singlet); 2.67 ? (1 H, septet, J= 7Hz)
13C NMR: 18.2, 27.2, 41.6, 211.2 ?
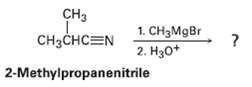
Transcribed Image Text:
CH3 CHзснCEN 1. CH3MGB 2. Нзо* 2-Methylpropanenitrile
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
The product is ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
(a) Unlike other esters, which react with Grignard reagents to give tertiary alcohols, ethyl Formate yields a different class of alcohols on treatment with Grignard reagents. What kind of alcohol is...
-
Epoxide?s react with Grignard reagents to yield alcohols propose a mechanism. 1. CHMgBr 2. * CH
-
Grignard reagents react slowly with oxetane to produce primary alcohols. Propose a mechanism for this reaction, and suggest why oxetane reacts with Grignard reagents even though most ethers do not....
-
Derive the transfer function of the circuit shown in figure below (for an ideal op amp) and show that it can be written in the form - V. -R2/R1 V," [1+ ( +j) Where w1 = 1/C,R1 and w2 = 1/ C2R2....
-
There is considerable disagreement as to whether government regulation has largely positive or negative influences on economic growth. Compare and contrast the ways in which government involvement in...
-
For a Y-connected load, the time-domain expressions for three line-to-neutral voltages at the terminals are: vAN = 150 cos ( t + 32) V vBN = 150 cos ( t - 88) V vCN = 150 cos ( t + 152) V Write the...
-
Using the chemical engineering literature, complete the detailed database for the detailed design of the base-case process in Figure 2.17. When appropriate, indicate the kind of data needed from a...
-
Neighbor Software Corp. has assembled the following data for the year ended December 31, 2010: Requirement 1. Prepare Neighbor Software Corp. s statement of cash flows using the indirect method to...
-
Data for Hermann Corporation are shown below: Selling price Variable expenses Contribution margin Per Unit Percent of Sales $ 110 77 100% 70 $ 33 30% Fixed expenses are $82,000 per month and the...
-
Sherina Smith (Social Security number 785-23-9873) lives at 536 West Lapham Street, Milwaukee, WI 53204, and is self-employed for 2019. She estimates her required annual estimated tax payment for...
-
Carboxylic acids (RCO 2 H) react with alcohols (R?OH) in the presence of an acid catalyst. The reaction product of propanoic acid with methanol has the following spectroscopic properties. Propose a...
-
Allene, H2C = C = CH2, has a heat of hydrogenation of 298kJ/mol (71.3 kcal/mol). Rank a conjugated diene, a non-conjugated diene, and an allene in order of stability.
-
Do you think the size of a firms market share or its conduct is the more reasonable basis for antitrust regulation? Explain your answer.
-
Given our focus on message processing, I believe it would be valuable to try applying the elaboration likelihood model (ELM) and/or the heuristic systematic model (HSM) to evaluate the goals of...
-
4 (B) On 1st January 2023, an investor has portfolio of Rs 1 Crore with a portfolio beta of 0.8. while the prices of nifty spot and nifty future are shown below: Stock Nifty spot Nifty July Futures...
-
Assume that the City of Coyote has produced its financial statements for December 31, 2024, and the year then ended. The city's general fund was only used to monitor education and parks. Its capital...
-
What is the most featured capability of Red-Black Tree? Answer: 3 7 10 11 18 22) 26 Black pointer Red pointer We have the next Red-Black Tree as seen above. Red pointers are shown as dashed while...
-
Use the sums 50 50 = 42,925 and k=1 (21 -86) - 0 k=1 50 k=1 k=1275 and the properties of summation to evaluate the given expression.
-
a. Express the signal in terms of unit-step functions. b. Find the Laplace transform of the expression in (a) by using the shift on \(t\)-axis. \(g(t)= 0 if t <0 t if 0 1
-
(a) Explain why the concentration of dissolved oxygen in freshwater is an important indicator of the quality of the water. (b) How is the solubility of oxygen in water affected by increasing...
-
Which molecule would you expect to be more soluble in water: CH 3 CH 2 CH 2 OH or HOCH 2 CH 2 CH 2 OH?
-
Ciprofloxacin is a member of the fluoroquinolone class of antibiotics. (a) Which of its rings are aromatic? (b) Which nitrogen atoms are basic? (c) Which protons would you expect to appear between 6...
-
Draw and name all the chlorinated benzenes having from one to six chlorine atoms.
-
Name the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) CH,CH,C CH OH CH CH CH NO COOH Br OCH(CH)2 OH NO NO CH,OCH,CH,
-
How much does the company pay in Dividends in 2023 when running the Base Case Drivers?
-
A portfolio has a standard deviation of 25%. The correlation of the portfolio and the market is 1. If the risk-free rate is 3.2%, the expected return on the market portfolio is 11%, and the standard...
-
(6 pts.) The following gas phase reaction system is conducted in an isothermal constant volume batch reactor. The reactor is charged with pure A with an initial concentration CAO AB ri = k CA B D r2...

Study smarter with the SolutionInn App


