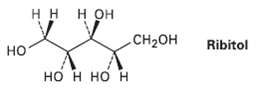
On catalytic hydrogenation over a platinum catalyst, ribose (Problem 9.57) is converted into ribitol. Is ribitol optically
Question:
On catalytic hydrogenation over a platinum catalyst, ribose (Problem 9.57) is converted into ribitol. Is ribitol optically active or inactive?Explain.
Transcribed Image Text:
нн нон CH2он Ribitol но но н но н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
Ribitol is an optically inactive meso compound Catalyti...View the full answer

Answered By

Sarfraz gull
have strong entrepreneurial and analytical skills which ensure quality tutoring and mentoring in your international business and management disciplines. Over last 3 years, I have expertise in the areas of Financial Planning, Business Management, Accounting, Finance, Corporate Finance, International Business, Human Resource Management, Entrepreneurship, Marketing, E-commerce, Social Media Marketing, and Supply Chain Management.
Over the years, I have been working as a business tutor and mentor for more than 3 years. Apart from tutoring online I have rich experience of working in multinational. I have worked on business management to project management.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
On catalytic hydrogenation over a rhodium catalyst, the compound shown gave a mixture containing cis-1-tert-butyl-4-methylcyclohexane (88%) and trans-1-tert-butyl-4 methylcyclohexane (12%). (a) What...
-
On catalytic hydrogenation over a rhodium catalyst, the compound shown gave a mixture containing cis-1-tert-butyl-4-methylcyclohexane (88%) and trans-1-tert-butyl-4-methylcyclohexane (12%). (a) What...
-
A triglyceride can be optically active if it contains two or more different fatty acids. (a) Draw the structure of an optically active triglyceride containing one equivalent of myristic acid and two...
-
Rand Medical manufactures lithotripters. Lithotripsy uses shock waves instead of surgery to eliminate kidney stones. Physicians' Leasing purchased a lithotripter from Rand for $2,000,000 and leased...
-
Is there a torque about the Moon's center of mass when the Moon's long axis is aligned with Earth's gravitational field? Explain how this compares with a magnetic compass.
-
Because of foundation settlement, a circular tower is leaning at an angle α to the vertical (see figure). The structural core of the tower is a circular cylinder of height h, outer...
-
Why might management not treat cyberthreats as a top priority?
-
Presented below are selected ledger accounts of Woods Corporation at December 31, 2012. Woods??s effective tax rate on all items is 34%. A physical inventory indicates that the ending inventory is...
-
How do I calculate the Portfolio Expected Return: You own aportfolio that has $4,600 invested in Stock X and $5,200 investedin Stock Z. What is the expected return on the portfolio if theexpected...
-
Jane and Rayna have asked for the income statement for JRS Inc. for the prior period from Shiva. In addition, they wanted additional information about the other manufacturing costs which increase...
-
Ribose, an essential part of ribonucleic acid (RNA), has the following structure: (a) How many chirality centers does ribose have? Identify them. (b) How many stereo isomers of ribose are there? (c)...
-
Hydroxylation of cis-2-hutene with OsO4 yields 2, 3-butanediol. What stereochemistry do you expect for the product?
-
How is it possible for an employee stock option to be valuable even if the firm's stock price fails to meet shareholders 'expectations?
-
Morgan Stanley analysts made the following forecasts for LinkedIn. (i) 26% CAGR revenue growth from 2011-2020. (ii) 35% EBITDA margin in 2020. (iii) Cost of capital of 11.5 % and (iv) terminal growth...
-
Jamar and Natasha had the following income and expenses during the year: $45,000 in wages. $24,000 in rental income. $8,000 in rental expenses. $48,000 in self-employment receipts. $12,000 in...
-
Seacrest Corporation sells sailboats and has an inventory turnover ratio of 2.45 times per year based on its most recent audited annual financial statements. Assume a 365 day year. During the year...
-
The alpha company produces and sells digital decoders and for the next six months the management has drawn up the following operating cost budget which refers to a sales forecast of 2,500 units of...
-
Jim McEntee is provided with a company car which he is allowed to use for private purposes. The car was purchased on 1 November 2017 for $40,000. During the year Jim drove the car 30,000 km of which...
-
The current exchange rate between the euro and the U.S. dollar is 0.925884. This means that one dollar can buy 0.925884 euros. How many dollars would you get for 1,000 euros?
-
Time Solutions, Inc. is an employment services firm that places both temporary and permanent workers with a variety of clients. Temporary placements account for 70% of Time Solutions' revenue;...
-
A solution contains one or more of the following ions: Hg 2 2 + , Ba 2 + , and Fe 2 + . When you add potassium chloride to the solution, a precipitate forms. The precipitate is filtered off, and you...
-
Planteose, a carbohydrate isolated from tobacco seeds, can be hydrolyzed in dilute acid to yield one equiva-lent each of D-fructose, D-glucose, and D-galactose. Almond emulsion (an enzyme preparation...
-
A process called sizing chemically modifies the cellulose in paper. As a result, the paper resists wetting (and thus prevent inks from running). In addition, sizing leaves the paper in a slightly...
-
Explain with a mechanism why treatment of the 2- deoxy-2-amino derivative of o-glucose (n-glucosamine) with aqueous NaOH liberates ammonia. HOCH HO OH NH2 D-glucosamine
-
What extent does emotional intelligence among leaders and employees contribute to effective teamwork and conflict resolution, and what methods can be employed to enhance emotional intelligence within...
-
Determine the exponential function y = c(b) that goes through the points (-2, 16) and (1,54). Algebraic work must be shown for full credit.
-
Solve 6. 8. 8 So log x (1 + x)(4+ x) [log x] 2 dx 0 1 + x So dx

Study smarter with the SolutionInn App


