Only one of the chlorines acts as a leaving group in this reactionexplain CI NaOH CI H2O
Question:
Only one of the chlorines acts as a leaving group in this reactionexplain
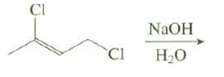
Transcribed Image Text:
CI NaOH CI H2O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
A chlorine attache...View the full answer

Answered By

Niala Orodi
I am a competent and an experienced writer with impeccable research and analytical skills. I am capable of producing quality content promptly. My core specialty includes health and medical sciences, but I can competently handle a vast majority of disciplines.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Only one of the following processes will occur measurably at room temperature. Which one? (a) F CI:
-
Explain why only one of the two chlorines of 1, 2-dichloro-2-methylpropane is replaced by a hydroxy group when the compound is heated in water (see the preceding hydrolysis reaction.
-
a) Only one of the two plaintiffs succeeded. Which one? b) Why did Maude's claim succeed? c) Why did Harold's claim fail?
-
Given that log (2) 0.91 and log (5) 2.1, evaluate each of the following. Hint: use the properties of logarithms to rewrite the given logarithm in terms of the the logarithms of 2 and 5. a) log(0.4)~...
-
A foundry employee worked a normal 38 hour week, but six hours were idle due to a small fire in the plant. The employee earns $29 per hour. Required: 1. Calculate the employee's total wages for the...
-
Daryl Kearns saved $240,000 during the 30 years that he worked for a major corporation. Now he has retired at the age of 60 and has begun to draw a comfortable pension check every month. He wants to...
-
Is there a best method for completing performance appraisals for each of the three different types of employees in an MNE? If so, describe the ways in which an HRIS may help.
-
Kelly, Gwen, and Tuoi incorporated their accounting business and own all its outstanding stock. During the current year, the corporation's taxable income is $300,000 after deducting salaries of...
-
Prospect Y = ($5, 0.25 ; $16, 0.75) If Will's utility of wealth function is given byu(x)=x^0.25, what is the value of CE(Y) for Will? (In other words, what is Will's certainty equivalent for prospect...
-
Find v such that p, q, u in Prob. 21 and v are in equilibrium. Data from Prob. 21 Find the resultant in terms of components and its magnitude. p = [2, 3, 0], q = [0, 6, 1], u = [2, 0, -4]
-
Explain why one of the oxygen's preferentially acts as the nucleophile in thisreaction:
-
This reaction gives two substitution products. Show the structures for these products and provide a mechanism for theirformation. . Br CH,
-
Figure P561 shows two designs for a beam to carry a repeated central load of 1200 lb. Which design would have the highest design factor for a given material? Data in Figure P561 10 in 8 in r = 0.20...
-
The local small appliance store has microwave ovens for sale at a price of \($149.95.\) The price tag notes that the manufacturers suggested retail price is \($160.\) This type of pricing is referred...
-
Why should a customer be concerned about transit inventory cost if they pay for the inventory only when the merchandise arrives at their premises?
-
Why do we say that transport is a derived demand?
-
Discuss whether a specific advertising campaign that promotes social equality by showing non-traditional families would turn you away from a product or make you consider using a product.
-
You just learned about new product development, branding, and pricing.Consider how this content relates to other areas of business. What information would you need to plan for how branding and...
-
For the system shown in Figure 5.46, the input is the force \(f\) and the outputs are the displacements \(x_{1}\) and \(x_{2}\) of the masses. a. Draw the necessary free-body diagrams and derive the...
-
On October 1, 2014, the Dow Jones Industrial Average (DJIA) opened at 17,042 points. During that day it lost 237 points. On October 2 it lost 4 points. On October 3 it gained 209 points. Deter-mine...
-
Sketch the graph of each line. 5x+ 4y=20 6543-2 1 9 # 3 2 H 2 m # 12 3 4 5 6 x
-
Draw and name all possible aromatic compounds with the formula C7H7C1.
-
Draw and name all possible aromatic Compounds with the formula C8H9Br.
-
Propose structures for aromatic hydrocarbons that meet the following descriptions: (a) C9H12; gives only one C9H11Br product on substitution with bromine (b) C10H14 gives only one C10H13C1 product on...
-
Security issues in today's workplace. Identify the different types of potential security issues an employer may have to deal with. Give examples to illustrate each of these potential security...
-
Research on the Internet and identify an emerging security issue. Describe the issue and its origin. Provide an example of this new security issue. Discuss the impacts of the security issue and...
-
A T-ball baseball player hits a baseball from a tee that is 0.6 m tall. The flight of the ball can be modelled by h(t) = -4.91 +61 +0.6, where h is the height in metres and t is the time in seconds....

Study smarter with the SolutionInn App


