Order each of the following sets of compounds with respect to SN1reactivity: H3C CH3 C NH2 (a)
Question:
Order each of the following sets of compounds with respect to SN1reactivity:
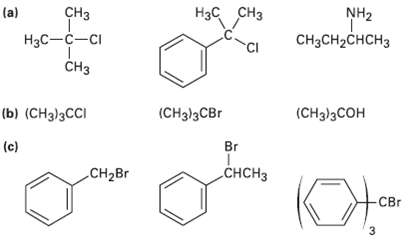
Transcribed Image Text:
H3C CH3 CНз NH2 (a) Нас—с—сi CH3CH2CHCH3 CI CHз (b) (CHз)3СCI (CHд)зСBr (CH)3CОН (c) Br CHCH3 Cн-Br CBr 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (16 reviews)
SN1 reactivity Least reactive a b c NH C...View the full answer

Answered By

Asd fgh
sadasmdna,smdna,smdna,msdn,masdn,masnd,masnd,m asd.as,dmas,dma.,sd as.dmas.,dma.,s ma.,sdm.,as mda.,smd.,asmd.,asmd.,asmd.,asm
5.00+
1+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Order each of the following sets of compounds with respect to SN2reactivity: CI CH (a) CH3CH2CHCH3 H CH3CH2CH2CI CH (b) CH CH CH CHCCH2Br CHH CH2Br Br CH (c) CH3CH2CH20CH3 CHCH2CH2Br CHCH2CH20Tos
-
Order each of the following sets from the least exothermic electron affinity to the most. a. F, Cl, Br, I b. N, O, F
-
Order the compounds in each of the following sets with respect to increasing acidity: (a) Acetic acid, oxalic acid, formic acid (b) p-Bromobenzoic acid, p-nitro benzoic acid, 2, 4-dinitrobenzoic acid...
-
Abardeen Corporation borrowed $90,000 from the bank on October 1, 2016. The note had an 8percent annual rate of interest and matured on March 31, 2017. Interest and principal were paid in cash on the...
-
How was it possible for a manufacturing plant in North America to have such a turnaround while in the recession?
-
1. Use descriptive statistics to summarize the data collected by Fresnos Manager of Product Quality for each of the 5 randomly selected dice. Based on these descriptive statistics, what are your...
-
Consider the situation of Example 12.10. Rather than shorting a futures contract, the U.S. firm could borrow \(500 /\left(1+r_{G} ight)\) euros (where \(r_{G}\) is the 90-day interest rate in...
-
A random sample of 174 college students was asked to indicate the number of hours per week that they surf the Internet for either personal information or material for a class assignment. The sample...
-
When Sergei's opened, it had to purchase most of its kitchen equipment. The equipment cost $ 4 0 , 0 0 0 new plus $ 5 , 0 0 0 to install. If food processing equipment is in the 1 0 years MACRS class,...
-
Gold Enterprises acquired 100 percent of Premium Builders stock on December 31, 20X4. Balance sheet data for Gold and Premium on January 1, 20X5, are as follows: At the date of the business...
-
The reactions shown below are unlikely to occur as written. Tell what is wrong with each, and predict the actualproduct. OCCH3)3 . H2CH Br (a) CHCH-CH (CH]l3c (b) Na* - CI LCH3 LCH3 (c) Socil,...
-
Predict the product and give the stereochemistry resulting from reaction of each of the following nucleophiles with (R)-2-bromooctane: (a) CN (b) CH3CO2 (c) CH3S
-
In Problems 53-56, show that the repeated application of L'Hopitals rule does not lead to a solution. Then use algebraic manipulation to evaluate each limit. 54. 56.
-
we can use the macroeconomic model to analyze the changes to real GDP (Y), unemployment (u) and price level (PL) as a result of some event. Draw the macroeconomic model at equilibrium.Label the axis...
-
Future of Taxation in the Philippines, if you were a policy maker or a legislator, what tax reform will you introduce and why? CONCEPT PAPER Proponent: Title: Proposed Tax Measure / Tax Reform...
-
Describe the role of population-level health in reducing/increasing the costs of healthcare at the individual level. Unlike other discussions, you do not need to comment on another student's post....
-
Identify and outline 5 rules that will be found in a collective bargaining agreement.
-
Discuss the Marxist perspective on employer relations and state how it's applicable to modern day workpalces.
-
Let \(X\) be a \(\mathrm{N}\left(\mu, \sigma^{2}ight)\) random variable. Using the moment generating function, derive the first three moments of \(X\). Repeat the process using the characteristic...
-
Archangel Corporation prepared the following variance report. Instructions Fill in the appropriate amounts or letters for the question marks in the report. ARCHANGEL CORPORATION Variance...
-
For the reaction 2 A(g) B(g) + 2 C(g), a reaction vessel initially contains only A at a pressure of P A = 255 mmHg. At equilibrium, P A = 55 mmHg. Calculate the value of K p .
-
Predict the major products formed when benzoyl chloride (PhCOCl) reacts with the following reagents. (a) Ethanol (b) Sodium acetate (c) Aniline (d) Anisole and aluminum chloride (e) Excess...
-
Predict the products of the following reactions. (a) Phenol + acetic anhydride (b) Phenol + acetic formic anhydride (c) Aniline + phthalic anhydride (d) Anisole + succinic anhydride and aluminum...
-
Acid-catalyzed transesterification and Fischer esterification take place by nearly identical mechanisms. Transesterification can also take place by a base-catalyzed mechanism, but all attempts at...
-
make a scenario for each of the following Gesture communication Facial expression communication Oral communication Written communication discuss briefly each scenario
-
Problem 7-30 Holding Period Yield (LO2) The YTM on a bond is the interest rate you earn on your investment if interest rates don't change. If you actually sell the bond before it matures, your...
-
According to theknot.com, online dating is the most common way people meet their spouses today. This claim was based on a survey of 1,400 randomly selected recently married couples, with 266...

Study smarter with the SolutionInn App


