Plot the vapour pressure data for a mixture of benzene (B) and acetic acid (A) given below
Question:
Plot the vapour pressure data for a mixture of benzene (B) and acetic acid (A) given below and plot the vapour pressure/composition curve for the mixture at 50°e. Then confirm that Raoult's and Henry's laws are obeyed in the appropriate regions. Deduce the activities and activity coefficients of the components on the Raoult's law basis and then, taking B as the solute,
its activity and activity coefficient on a Henry's law basis. Finally,
evaluate the excess Gibbs energy of the mixture over the composition range spanned by the data.
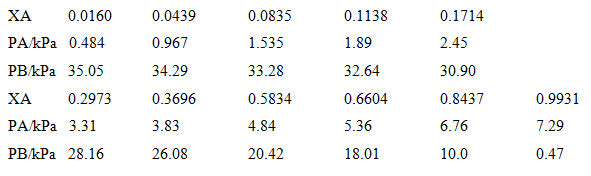
Transcribed Image Text:
0.0160 0.0439 0.0835 0.1714 XA 0.1138 2.45 PAKPA 0.484 1.535 1.89 0.967 34.29 33.28 PB/kPa 35.05 32.64 0.6604 30.90 0.8437 0.2973 0.3696 0.5834 XA 0.9931 PAKPA 3.31 5.36 7.29 3.83 4.84 6.76 PB/kPa 28.16 26.08 20.42 18.01 0.47 10.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
The data are plotted in Figure 53 The regions where the vapor pressure curves show approximate strai...View the full answer

Answered By

Akash Deshmukh
My name is Akash Deshmukh . I'm pursuing masters in statistics. I have adequate knowledge about Statistics. My sheer dedication and interest towards statistics can be helpful for Students. From last 1 year I'm working as subject matter expert where I can solve students doubt about there study as well I help them to guide in there future career I always interested to teach new things to students. Since couple of years I been practicing Python, R programming , MySQL, tableau , Advanced Excel and know Machine learning tools and techniques very well like regression, classification, NLP, statistical modeling, clustering, image recognition, object detection ,recommendation system and many more.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physical Chemistry questions
-
In Example 11.6, a mixture of benzene and cyclohexane is separated in a separation sequence that begins with homogeneous azeotropic distillation using acetone as the entrainer. Can the same...
-
A mixture of benzene and dichloroethane is used to test the efficiency of a packed column that contains 10 ft of packing and operates adiabatically at atmospheric pressure. The liquid is charged to...
-
For a mixture of saturated vapor and saturated liquid of water at 200oC, use tabulated properties or the PC system state TESTcalc to show that the specific Gibbs functions of the two phases are equal.
-
1. As a policy maker you should never worry much about those are eligible for Medicaid benefits and do not enroll. This is because they will enroll in public insurance if they need it. True or False?...
-
(a) What is the advantage of having a cerebrum that is highly convoluted? (b) What term is used to indicate its grooves? Its outward folds? (c) Which groove divides the cerebrum into two hemispheres?...
-
1. In a partnership liquidation, the final cash distribution to the partners should be made in accordance with the: a. Partner profit and loss sharing ratios b. Balances of partner capital accounts...
-
Calculate the kinetic energy correction factor for each of the following velocity profiles for a circular pipe: (a) \(u=u_{\max }\left(1-\frac{r}{R} ight)\) (b) \(u=u_{\max...
-
Farrah Westin plans to build a concrete walkway for her home during her vacation. The following schedule shows how project time will be allocated: Hours Purchase materials ............5 Obtain rental...
-
Let $4 million is invested in shares in ABC Ltd, which have an estimated return of zero and a standard deviation of 30 per cent per annum. Also assume that $7 million is invested in shares in XYZ...
-
Pams Creations had the following sales and purchase transactions during 2016. Beginning inventory consisted of 60 items at $350 each. The company uses the FIFO cost flow assumption and keeps...
-
For a first-order phase transition, to which the Clapeyron equation does apply, prove the relation C3 = CP - aVurH/urs V Where Cs = (q/T) s is the heat capacity along the coexistence curve of two...
-
Comelli and Francesconi examined mixtures of propionic acid with various other organic liquids at 313.15 K (F. Comelli and R. Francesconi, Chem. Eng. Data 41,101 (1996)). They report the excess...
-
In the August 18, 1989, issue of the Will Street Journal there appeared an article entitled "Program Trading Spreads from Just Wall Street Firms." Following are two quotations from that article:...
-
The economy is absorbing excess capacity more rapidly than we projected in April, and it now appears that the output gap will close around the end of this year. . . . Meanwhile, inflation has...
-
On the graph, show the long-run effects of the pollution crackdown. California plans to crack down on the use of fumigants by growers of strawberries. The biggest burden will fall on Ventura Countys...
-
Chromium(VI) forms two different oxyanions, the orange dichromate ion (Cr 2 O 7 2 )and the yellow chromate ion (CrO 4 2 ) Extra \left or missing ight . The equilibrium reaction between the two ions...
-
Three diatomic molecules from different gas samplesone at \(3 \mathrm{~K}\), one at \(298 \mathrm{~K}\), and one at \(1000 \mathrm{~K}\)-are in thermal equilibrium with their respective surroundings....
-
Suppose a glass rod is rubbed on wool. When held \(35.0 \mathrm{~mm}\) above a \(0.20 \mathrm{~g}\) scrap of paper, the rod lifts the paper with an initial acceleration of \(0.14 \mathrm{~m} /...
-
April and Andy were married on May 16, 1998. They had two children. Noah Andrew was born on July 19, 1999. Emma Katherine was born on July 30, 2000 . . . In the spring of 2003, April and Andy...
-
A routine activity such as pumping gasoline can be related to many of the concepts studied in this text. Suppose that premium unleaded costs $3.75 per gal. Work Exercises in order. Use the...
-
Indicate how important each characteristic is to you. Answer according to your feelings about the most recent job you had or about the job you currently hold. Circle the number on the scale that...
-
Explain the origin of the symmetry number.
-
Explore whether a magnetic field can influence the heat capacity of a paramagnetic molecule by calculating the electronic contribution to the heat capacity of an NO 2 molecule in a magnetic field....
-
Consider Stirlings approximation for ln N! in the derivation of the Boltzmann distribution. What difference would it make if (a) A cruder approximation, N! = N N , (b) The better approximation in...
-
Large-scale industrial production - accompanied by massive technological change, expanding international communication networks, and pro-growth government policies-generated rapid economic...
-
How can this relate to individuals who have had a TBI and are currently homeless or marginally housed?
-
1. According to Marxian theory, are crises an economic process inherent to the accumulation dynamics of current capitalism? Justify your answer. 2. According to Marxian theory, what do you consider...

Study smarter with the SolutionInn App


