Predict the major product(s) of the followingreactions: (b) (a) CH3CH2COCI AICI3 CH3CH2CI AICI3 (c) (d) N(CH2CH3}2 CO2H
Question:
Predict the major product(s) of the followingreactions:
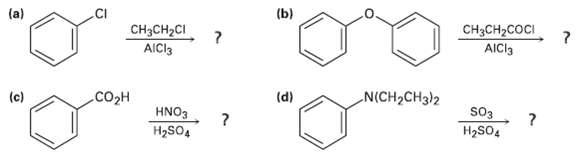
Transcribed Image Text:
(b) (a) CH3CH2COCI AICI3 CH3CH2CI AICI3 (c) (d) N(CH2CH3}2 CO2H HNO3 so3 H2SO4 H2SO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
b c CH3CHCl AICI 3 COH ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the major product (or products) that would be obtained when each of the following compounds is nitrated: (a) (b) (c) OH CF CN SO3H OCH3 NO2
-
Predict the major product for each proposed Diels-Alder reaction. Include stereochemistry where appropriate. (a) (b) (c) Ph 0 Ph
-
Predict the major product formed when 2-bromobutane is subjected to dehydrobromination using sodium ethoxide in ethanol at 55oC.
-
Review TP1. Review current season ticket prices for one Major League Baseball team. Choose one season ticket price area to review. A. Determine what is recognized as per ticket revenue after each...
-
During 2016, John was the chief executive officer and a shareholder of Maze, Inc. He owned 60% of the outstanding stock of Maze. In 2013, John and Maze, as co-borrowers, obtained a $100,000 loan from...
-
Here is a student procedure to measure nicotine in urine. A 1.00-mL sample of biological fluid was placed in a 12-mL vial containing 0.7 g Na2CO3 powder. After 5.00 g of the internal standard...
-
P(0 < z < 2.315) Find the indicated probability using the standard normal distribution. If convenient, use technology to find the probability.
-
R. J. Winter Co. recently issued $100,000, 10-year deferred interest bonds. The bonds have a stated rate of 10%, and interest is to be paid in 10 semiannual payments beginning in Year 6. The market...
-
(c) Consider three scenarios: Base Case Worst Case Best Case % of Members Who Do Not Show 25% 50% 15% % of Nonmembers Who Do Not Show Number of Nonmember Registrants 10% 130 30% 100 5% 150 All other...
-
Use the data in MEAP00_01.RAW to answer this question. (i) Estimate the model mathA = (0 + (1 lunch + (2 log(enroll) + (3log(exppp) + u by OLS and obtain the usual standard errors and the fully...
-
What product(s) would you expect to obtain from the followingreactions? (b) r. 1. HNO3, H2SO4 2. Fe, H30* "C Br H2/Pd NO2 (d) CI. (c) CH3CH2CH2CI MO 2 AICI3
-
Aromatic iodination can be carried out with a number of reagents, including iodine mono chloride, ICI. What is the direction of polarization of ICI? Propose a mechanism for the iodination of an...
-
Papadopoulos Limited (PL) sells retail merchandise in Canada. The company was incorporated last year and is now in its second year of operations. PL is owned and operated by the Papadopoulos family,...
-
If x= sin2t, y = cos2t, are parametric equations. find dy dx
-
What major factors affect Domino's or any other major company in dealing in global markets?
-
In order to obtain a good whose Engel curve is hump-shaped (i.e., a good that is initially normal but later inferior as income increases), we must have preferences that are not well-behaved (i.e.,...
-
Given the following circuit with resistors R = 1.00, R = 2.00, R3 = 3.00, R = 4.00, Rs = 5.0 and R = 6.00, with E = 10.0V, E3 = 10.0V, and Es = 15.0V. (a) What is the current /2 passing through...
-
What type of HR supply model utilizes a series of matrices that detail the various patterns of movement to and from the various jobs in an organization?
-
Which of the following asset allocation methods would not likely be used by Nowacki and Knight to select investments for the existing equity fund? A. Sector and industry rotation B. Growth at a...
-
d) For die casting processes: 1. What are the most common metals processed using die casting and discuss why other metals are not commonly die casted? 2. Which die casting machines usually have a...
-
Two separate experiments are performed on a gas enclosed in a piston-and-cylinder device, both starting from the same initial state. The result of the first experiment is to be used to predict the...
-
Show all the steps in the mechanisms for these reactions. Don't forgot to use curved arrows to show the movement of electrons in eachstep. CH3 CH3 ) . + CHH ., + Br CH; CH3 H,SO, CH;CH,OCH,CH; + H;O...
-
Explain how both enantiomers of the product are formed in the reaction shown in problem 10.42c.
-
Show all the steps in the mechanism for thisreaction: CH,Br CH2OET ELOH OEt
-
- Let [121] A-03 1 4 12 If possible, find a matrix C such that 1 2 AC=01 41
-
Provide References In September of 2015, the EPA issued Volkswagen ( OTCPK:VLKAY ) a notice of violation of the Clean Air Act of 1963. The EPA accused the Volkswagen Group of intentionally...
-
2. Find c such that A is invertible 1 0 A= -1 -1 3 1 0 2c -4]

Study smarter with the SolutionInn App


