Predict the product from reaction of the following substance with: (a) NaBH4 then H3O+ (b) LiAlH4 then
Question:
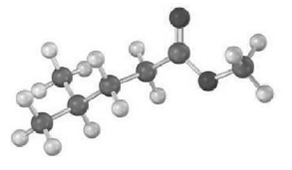
Predict the product from reaction of the following substance with:
(a) NaBH4 then H3O+
(b) LiAlH4 then H3O+
(c) CH3CH2 MgBr; then H3O+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
a b c CH3 i CH3CHCHCHCOCH 3 CH3 CH3C...View the full answer

Answered By

RADHIKA MEENAKAR
I am a qualified indian Company Secretary along with Masters in finance with over 6 plus years of professional experience. Apart from this i am a certified accounts and finance tutor on many online platforms.
My Linkedin profile link is here https://www.linkedin.com/in/radhika-meenakar-88b9808a/
5.00+
12+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the product from reaction of the following substance (reddish brown = Br) with: (a) PBr3 (b) Aqueous H2SO4 (c) SOCl2 (d) PCC (e) Br2,FeBr3
-
Predict the product from the SN2 reaction of a. Trans-4-methylcyclohexyl bromide with cyanide ion. b. (S)-2-bromopentane with cyanide ion. c. (R)-2-chlorobutane with NaSH.
-
Predict the product from the reaction of phenylmagnesium bromide (C6H5MgBr) with benzoyl chloride (C6H5COCl).
-
Melissa is a sole trader. Her capital gains and capital losses for 2020-21 are 27,000 and 700 respectively. She has capital losses brought forward from 2019-20 of 12,900 and she also has unrelieved...
-
What are three ways a firm can incorporate the advantage of speed in its business? Use Exhibit 8.6 to help you answer this question. Can you give an example of a company that has done this?
-
What test can be used to assess whether the mean change in BMI over 6 years is different between women who quit smoking and women who have never smoked? One issue is that there has been a secular...
-
An experiment has the four possible mutually exclusive outcomes \(A, B, C\), and \(D\). Check whether the following assignments of probability are permissible: (a) \(P(A)=0.38, P(B)=0.16, P(C)=0.11,...
-
Use the Access_Practice.accdb database to complete the following tasks in Access. a. The database contains three tables containing information about this companys sales process: Inventory, Sales, and...
-
A girl delivering newspapers covers her route by traveling 8.70 blocks north, 2.18 blocks south, and 9.93 blocks east. Find the angle (a positive value in , between 0 and 360) of her resultant...
-
Bug-Off Exterminators provides pest control services and sells extermination products manufactured by other companies. The following six-column table contains the company's unadjusted trial balance...
-
Draw the structure of the carbonyl compound(s) from which each of the following alcohols might have been prepared, and show the products you would obtain by treatment of each alcohol with (i) Na...
-
Name and assign R or S stereochemistry to the product(s) you would obtain by reaction of the following substance with ethyl magnesium bromide. Is the product chiral? Is it optically active? Explain.
-
On January 1, 2012, Picante Corporation acquired 100 percent of the outstanding voting stock of Salsa Corporation for $1,765,000 cash. On the acquisition date, Salsa had the following balance sheet:...
-
Submit a short memo to your instructor explaining special and extraordinary expenses and how they are calculated? . The Family Law in British Columbia site is, again, a good resource. Cite all of...
-
With reference to decided cases and other authorities, compare and contrast how the following unions are dealt with in the Namibian and South African legal systems: 1.1 Civil Unions 1.2 Co-Habitation...
-
Hanna works as a teller a bank. One of Hanna's clients, an out of province student, started to consistently deposit large amounts of cash in the form of $5, $10, and $20 Bills. Hanna asked her client...
-
In a chem lab, you have in stock a 10% saline solution and a 50% saline solution. However, you need to create 500 milliliters (mL) of a 40% saline solution. How much of each should you mix to obtain...
-
What role does nonverbal communication, including body language, facial expressions, and paralinguistic cues, play in shaping interpersonal interactions, influencing perceptions, and conveying...
-
Does your state impose the death penalty? If so, what does the state law provide with respect to juries considering aggravating and mitigating factors? Do you think your states law governing the...
-
Describe a group you belong or have belonged discuss the stages of group development and suggest how to improve the group effectiveness by using the group development model.
-
The normal boiling point of acetone is 56.2 C, and the molar heat of vaporization is 32.0 kJ mol 1 . What is the boiling temperature of acetone under a pressure of 50.0 mmHg?
-
An alkane with the formula C6H14 can be prepared by hydrogenation of either of only two precursor alkenes having the formula C6H12. Write the structure of this alkane, give its IUPAC name, and show...
-
Rank the following compounds in order of increasing stability based on relative ring strain.
-
Write the structures of two chair conformations of 1-tert-butyl-1-methylcyclohexane. Which conformation is more stable? Explain your answer.
-
How might technology change in the future? How will technology make our lives better in the next ten years? What kinds of technology are you most excited about? What do you think will be the next...
-
How do the local electronic and printing advertisements apply in the real ground? What are the most accepted brand names? What are the good qualities and demerits of the brand?
-
Gary and Linda assume they will need $70,000 at the beginning of each year in today's dollars when they retire in 6 years. The couple is assuming an annual inflation rate of 4% and a 7.5% after-tax...

Study smarter with the SolutionInn App


