Predict the products of the following nucleophilic acyl substitutionreactions: (b) (a) NaOH NH3 H20 H
Question:
Predict the products of the following nucleophilic acyl substitutionreactions:
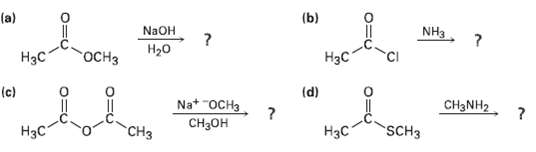
Transcribed Image Text:
(b) (a) NaOH NH3 H20 оСHз Нас Нэс (d) (c) Na* "OCH3 Cнзон CH3NH2 SCH3 Нзс "CHз НаС
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Strategy Identify the nucleophile boxed and the leavin...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the mechanism of the following nucleophilic acyl substitution reaction, using curved arrows to indicate the electron flow in eachstep: Nat -H CH
-
Predict the products of the following reactions: (a) Excess NH3 + Ph - CH2CH2CH2Br (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) (o) (p) (q) (r) (I) NaN3 (2) LiAIH (3) H30 1-bromopentane CH3...
-
Predict the products of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) OH CI CH NH2 Ph-C-CI+ NH2 0 + (D LiAIH (2) H20 (2) H,0 -. O (I) excess PhMgBr (2) H,o ( CH Mgl (2)...
-
In five sentences or more answer the following question: Was Japanese Internment necessary to protect Americas national security? Why / why not?
-
What can you do to appear professional when a potential employer contacts you by phone for a screening interview or to schedule a job interview?
-
The model in Section 5.5, Exercise 35. Find the null clines and equilibria of this model when = 2.0, = 1.0, and k = 1.0. Draw the null clines and find equilibria of the above extensions of the...
-
What are the inputs and the steps in applying earned value management as a project management tool?
-
Which of the following products and services are likely to encounter adverse selection problems: golf shirts at traveling pro tournaments, certified gemstones from Tiffanys, graduation gift travel...
-
The COVID-19 pandemic continues to challenge governments and policymakers worldwide. They have rightfully prioritised reducing the spread of the virus through social distancing interventions....
-
Stockholders equity totaled $246,000 at the beginning of the year. During the year, net income was $36,000, dividends of $9,000 were declared and paid, and $30,000 of common stock was issued at par...
-
Rank the compounds in each of the following sets in order of their expected reactivity toward nucleophilic acylsubstitution: (a) , , , CH, HH. NH2 (b) CHC -CCI3, CHCICF3/2
-
The following structure represents tetrahedral alkoxide ion intermediate formed by addition of a nucleophile to a carboxylic acid derivative. Identify the nucleophile, the leaving group, the starting...
-
Describe the effect of a change in each of the following factors on the value of a call option: i. Stock price ii. Exercise price iii. Option life iv. Risk-free rate v. Stock price variance (that is,...
-
Leach Finishing makes various metal fittings for the construction Industry. Three of the fittings, models X-12, X-24, and X-30, require grinding on a patented machine of which Leach has only one. The...
-
Jurvin Enterprises is a manufacturing company with no beginning inventories. A subset of the transactions it recorded during a recent month is shown below. a. Purchased $94,000 in raw materials for...
-
Sheffield manufactures a fine wine in two departments, Fermenting and Bottling. In the Fermenting Department, grapes are aged in casks for a period of 30 days. In the Bottling Department, the wine is...
-
Larned Corporation recorded the following transactions for the just completed month. a. Purchased $80,000 of raw materials on account. b. $71,000 in raw materials were used in production. Of this...
-
Raner, Harris and Chan is a consulting firm that specializes in information systems for medical and dental clinics. The firm has two offices-one in Chicago and one in Minneapolis. The firm classifies...
-
Why is the inner core Earths most dense region? (a) The inner core is made of mostly iron, which is very dense. (b) Dense material sank toward Earths center when Earth was molten. (c) Pressure packs...
-
Find a polar equation for the curve represented by the given Cartesian equation. 4y 2 = x
-
The structure of acetylsalicylic acid (aspirin) is shown here. How many pi bonds are present in acetylsalicylic acid? How many sigma bonds? What parts of the molecule are free to rotate? What parts...
-
Explain why a mixture of two isomeric ethers is formed in the following reaction. NaBH
-
Predict the absolute configuration of the major diol product formed by treatment of (S)-2 ethyl- 2-methyloxirane with water in the presence of an acid catalyst.
-
When (3s,4S)-4-methoxy-3-methyl- I -pentene is treated with mercuric acetate in methanol solvent, then with NaBH4, two isomeric compounds with the formula C8H18O2, are isolated. One, compound A, is...
-
A drum contains water and 1,4-dioxane that is left over waste from an electronics cleaning facility. For a mixture of 70 mol% water and 30 mol% 1,4-dioxane in the drum at 35 oC, calculate the...
-
Show what is produced by the following Java code. Assume list is a reference variable of type Sinly LinkedList, and length(), InsertFirst(), insertLast). first(), last(). print() are the methods of...
-
Explore the themes of alienation and dislocation in James Joyce's "Ulysses." How do these themes reflect the modernist perspective on the fragmentation of society and the individual's place within it...

Study smarter with the SolutionInn App


