Predict the product(s) of the following reactions: CH (b) CH-CH (a) 1. DIBAH 2. H30+ 1. CH2CH,MgBr
Question:
Predict the product(s) of the following reactions:
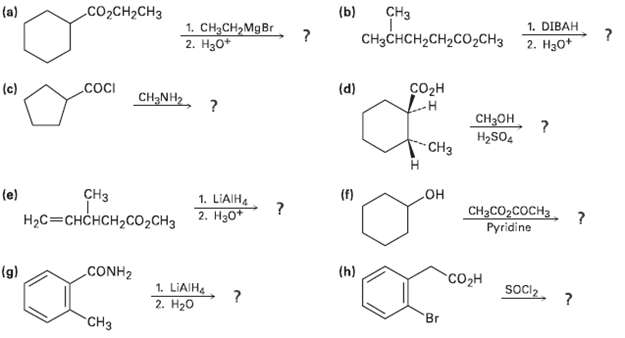
Transcribed Image Text:
CHз (b) СооCH-CHз (a) 1. DIBAH 2. H30+ 1. CH2CH,MgBr 2. Нао" CHзснCH-CH2CO2CH3 (d) Созн .cocI (c) CH NH2, ? CH3он H2SO4 "CHз но CH3CO2COCH3 (f) СНз 1. LIAIH (e) 2. Нзо" Pyridine НаС3 СHCHCH>со,сна (h) "сон CONH2 (g) Socl2. 1. LIAIH. 2. H20 Br "CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
a b c O0 OCHCH3 1 CH3CHMgBr 2 H30 CH3 CH3CHCHCHCOCH 3 i The 1 DIBA...View the full answer

Answered By

Munibah Munir
I've done MS specialization in finance’s have command on accounting and financial management. Forecasting and Financial Statement Analysis is basic field of my specialization. On many firms I have done real base projects in financial management field special forecasting. I have served more than 500 Clients for more than 800 business projects, and I have got a very high repute in providing highly professional and quality services.I have capability of performing extra-ordinarily well in limited time and at reasonable fee. My clients are guaranteed full satisfaction and I make things easy for them. I am capable of handling complex issues in the mentioned areas and never let my clients down.
4.60+
467+ Reviews
648+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the product of each of the following Diels-Alder reactions for the diene (left) and dienophile (right) as shown below. 0 0 0 0
-
Predict whether the following reactions will be spontaneous in acidic solution under standard conditions: (a) Oxidation of Sn to Sn2+ by I2 (to form I-) (b) Reduction of Ni2+ to Ni by I- (to form I2)...
-
Predict whether the following reactions would occur spontaneously in aqueous solution at 25C. Assume that the initial concentrations of dissolved species are all 1.0 M. (a) Ca(s) + Cd2+(aq) Ca2+(aq)...
-
A man drops a stone from a high bridge and hears it strike the water below exactly 4 s later. (a) Estimate the distance to the water based on the assumption that the travel time for the sound to...
-
Why should even practiced speakers plan their presentations when addressing a business audience instead of just "winging it"?
-
During a 10-minute interval, adult worms switch from eating to egg-laying with probability 0.1 and from egg-laying to eating with probability 0.15. a. Draw a diagram illustrating this process. b....
-
Create a simple WBS for planning a wedding.
-
Ben Toucan, owner of the Aspen Restaurant, wants to determine the present value of his investment. The Aspen Restaurant is currently in the development stage but Toucan hopes to begin operations...
-
The terms below are just a few terms needed in discussing the world of social media marketing and it is important that students have a clear and consistent understanding of these terms. We will use...
-
Hospital personnel routinely examine patient records for error, such as incomplete insurance information, on incomplete patient history, or missing/incomplete medical records. On average, about 250...
-
How might you prepare the following compounds from butanoic acid? (a) 1-Bulanol (b) Butanal (c) 1-Bromobutane (d) Pentanenitrile (e) 1-Butene (f) N-Methylpentanamidc (g) 2-Hexanone (h) Butyl benzene...
-
Predict the product, if any, of reaction between propanoyl chloride and the following reagents: (a) Li (Ph) 2 Cu in ether (b) LiA1H 4 , then H 3 O + (c) CH 3 MgBr, then H 3 O + (d) H 3 O + (e)...
-
How does primary succession differ from secondary succession?
-
Question: Part One: Memo Write an analysis of the impact of tax laws and incentives as demonstrated in this current years tax return. Specifically, you must address the following: Explain the...
-
Question: Tania, Inc. issued a note payable with principal and interest due at the end of the term of the note. Details about the note payable are as follows:Issuance date: October 1, fear...
-
Question: A company had the following purchases and sales during the month of November:\table[[Date,Activities,Units Acquired at Cost,Units Sold at Retail],[November 1,Beginning inventory,5 units @...
-
Question: d. ?During December, Ingrid Legal Services provided legal services, and the client prepaid $7,000. ?Ingrid Legal Services recorded this amount as Unearned Revenue. The job will take several...
-
George is a street vendor of chicken wings. He offers them in mild, medium, and flaming varieties, but most customers preter the hottest option. Typically, his sales are comprised of 2 0 % ?mild, 2 0...
-
What does the layer of iridium in the rock record tell about the demise of dinosaurs?
-
A spacecraft has left the earth and is moving toward Mars. An observer on the earth finds that, relative to measurements made when the spacecraft was at rest, its a. length is shorter b. KE is less...
-
The valence electron configurations of several atoms are shown here. How many bonds can each atom make without hybridization? a. Be 2s 2 b. P 3s 2 3p 3 c. F 2s 2 2p 5
-
The 60-MHz proton NMR spectrum of 2, 2, 3, 3-tetra-chlorobutane consists of a sharp singlet at 25oC, but at -45oC consists of two singlets of different intensities separated by about 10 Hz. Explain...
-
The 60-MHz proton NMR spectrum of 2, 2, 3, 3-tetra-chlorobutane consists of a sharp singlet at 25oC, but at -45oC consists of two singlets of different intensities separated by about 10 Hz. Explain...
-
What changes would you expect in the 13C NMR spectrum of 1-bromopropane upon cooling the compound to very low temperature?
-
The following account balances are taken from the ledger of Maxwell Limited on 31 December 2018, the end of its fiscal year: Maxwell Limited Trial Balance As on 31 December 2018 S'm S'm Investment...
-
A researcher wishes to assess whether vitamin C is effective in the treatment of colds. To evaluate this hypothesis, the researcher decides to conduct a 2-year experimental study. The researcher...
-
Cullumber Co. acquired 25% of the 520,000 shares of outstanding common stock of Oriole Inc. on December 31, 2025. The purchase price was $3,913,000. Oriole declared and paid $1.20 per share cash...

Study smarter with the SolutionInn App


