Predict the products of the following reactions: CH (b) CH (a) 1. (NH2)2C=S 2. NaOH, H20 -CH
Question:
Predict the products of the following reactions:
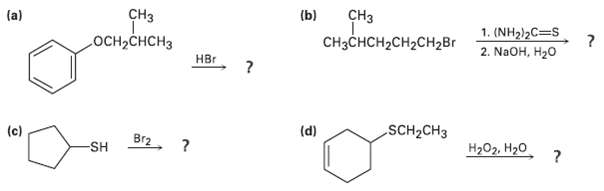
Transcribed Image Text:
CHз (b) CHз (a) 1. (NH2)2C=S 2. NaOH, H20 осн-снCHз CнзснсH2CH2CH2Br Hвr SCH2CH3 (d) (c) Br2, ? Hа0z. Н2о SH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
b O d CH3 OCHCHCH3 CH3 CH3...View the full answer

Answered By

Sumit kumar
I am an experienced online essay writer with a thorough understanding of any curriculum.and subject expert at Chegg for mathematics, CS subjects..
4.90+
5+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry when relevant. H2/Pd (a) Br2 (b) Os04 (c) NMO Cl2, H20 (d) CH212, Zn/Cu (e)...
-
Predict the products of the following reactions: (a) (b) CH CH3CH2CH-0-CH2CH2CH3 CH r HBr
-
Predict the products of the following reactions. When more than one product is expected, predict which will be the major product. (a) (b) (c) (d) OH H2SO4 heat H3PO4 heat BrNaocH, CH CH H,SO4 heat OH
-
To load and register a JDBC driver, two popular methods can be used: either using the Class.forName() method or creating a new instance of the Driver class.(True/False)
-
What issues do they encounter when attempting to measure the economy?
-
At first glance, the structure of diborane would seem unusual. Why shouldnt the molecule assume the same geometry as ethane, which after all has the same number of heavy atoms and the same number of...
-
Price Differentiating E-commerce websites "alter results depending on whether consumers use smartphones or particular web browsers," 34 reports a new study. The researchers created clean accounts...
-
Tyler Pahl recently received the following information related to Pahl Companys December 31, 2014, balance sheet. Inventory ................$ 4,100 Cash .................. 3,900 Equipment...
-
Toss a fair coin twice. You win $4 if both one Head and one Tails appears, lose $4 if no toss comes out Heads, and win or lose nothing otherwise. (a) What is the expected number of games you need to...
-
Calculate the linear convolution and circular convolution of the following sequence: x[k] = k(u[k] - u[k - 3]) {h[k]} = {1, 2, 1, }
-
How would you prepare the following compounds from 1-phenylethanol? (a) Methyl 1-phenyl ethyl ether (b) Phenylepoxyethane (c) tert-Butyl 1-phenylethyl ether (d) 1-Phenylethanethiol
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
Ascorbic acid (Vitamin C) has the empirical formula of C 3 H 4 O 3 and a molar mass of 176.13 g/mol. Find its molecular formula.
-
Describe the characteristics of an effective fee agreement and identify the various types of fee agreements that may be used in divorce cases. List the kinds of resources included in a paralegal's...
-
What are the regulatory agency changes coming in the near future?
-
Explain the reasons why law reform for domestic violence has been effective in achieving justice for different groups in society. Refer to the Family Law Act 1975 and the legislation Family Law Act...
-
If the mean absolute percentage error (MAPE) is at a -4% what should the hotel implement immediately? Explain.
-
An insurance agent is trying to sell you an immediate retirement annuity that offers $16,000 per year at the end of each of the next 30 years. The price of the investment proposed by the agent is...
-
In 2006 in Hudson v. Michigan, Justice Scalia, writing for a sharply divided Supreme Court, concluded that when it comes to knock-and-announce violations, [r]esort to the massive remedy of...
-
Q:1 Take any product or service offered in Pakistan and apply all determinents of customer Perceived value ?
-
It is necessary to estimate how rapidly a piece of equipment can be evacuated. The equipment, which is 0.7 m 3 in volume, initially contains carbon dioxide at 340 K and 1 bar pressure. The equipment...
-
Assign the proper formal charge to the colored atom in each of the following structures: (a) (b) (c) (d) (e) (f) (g) (h) H-C-H H-C-NH H-C-H
-
Write a condensed structural formula for the following compound.
-
Write each of the following condensed structural formulas as a bond-line formula: (a) (CH3)2CHCH2CH3 (b) (CH3)2CHCH2CH2OH (c) (CH3)2C==CHCH2CH3 (d) CH3CH2CH2CH2CH3 (e) CH3CH2CH(OH)CH2CH3 (f) CH2...
-
Describe the value chain analysis methods that a firm might utilize to identify strategies to enhance organizational capabilities.
-
Nurses are advocates for patients and must find a balance while delivering patient care. Discuss
-
The geriatric nurse cares for patients who are aging or suffering from conditions normally, Discuss?

Study smarter with the SolutionInn App


