Predict whether each of the following substitution reactions is likely to be SN1 orSN2. (a) HCI
Question:
Predict whether each of the following substitution reactions is likely to be SN1 orSN2.
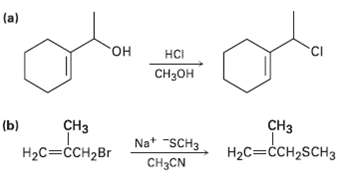
Transcribed Image Text:
(a) он HCI Cнзон сHз CHз (b) Na* "SCH3 H2C=ĊCH2SCH3 Нас%3ссH2Br CH3CN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
s Strategy Both substrates have allylic groups and m...View the full answer

Answered By

Michael Owens
I am a competent Software Engineer with sufficient experience in web applications development using the following programming languages:-
HTML5, CSS3, PHP, JAVASCRIPT, TYPESCRIPT AND SQL.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict whether each of the following carbocations will rearrange. If so, draw the expected rearrangement using curved arrows. a. b. c. d. e. f. g.
-
Predict whether each of the following elements will act as a donor or an acceptor when added to the indicated semiconducting material. Assume that the impurity elements are substitutional. Tmurity...
-
Each of the following nucleophilic substitution reactions has been reported in the chemical literature. Many of them involve reactants that are somewhat more complex than those we have dealt with to...
-
Dickletton Attorneys' policy is to bank all receipts in its Trust bank account and at the end of each month the bookkeeper transfers the relevant funds due to Dickletton Attorneys from the trust to...
-
Within months of Barra's appointment as CEO and during the time of the internal investigation, several executives departed the company. Does this suggest that GM was managing turnover effectively?...
-
The acetylacetone ion forms very stable complexes with many metallic ions. It acts as a bidentate ligand, coordinating to the metal at two adjacent positions. Suppose that one of the CH3 groups of...
-
Following are several tests of controls introduced in the chapter: Examine periodic reports by non-Purchasing Department personnel regarding purchase prices and practices. Observe Receiving...
-
Why did lagging depreciation legislation increase the importance of using capital funds optimally? MINI CASE Heublein, Inc., develops, manufactures, and markets consumer food and beverage products...
-
Bob the Builder and Wendy were partners in their constructionbusiness and were general contractors. They submitted a bid toconstruct a new building for the the city. They includedLumberjack\'s quote...
-
A vaccine to prevent severe rotavirus gastroenteritis (diarrhea) was given to African children within the first year of life as part of a drug study. The study reported that of the 3298 children...
-
3-Bromo-1-butene and 1-bromo-2-butene undergo SN1 reaction at nearly the same rate even though one is a secondary halide and the other is primary. Explain
-
Review the mechanism of geraniol biosynthesis shown in Figure and then propose a mechanism for the biosynthesis of limonene from linalyldiphosphate. OPP Linalyl diphosphate Limonene
-
Auto manufacturers buy car components from suppliers who deliver them to the assembly line. A manufacturer can schedule staff time to check a sample of 5 parts a day for defects or a sample of 25...
-
The Cubby Bear started the month with a food inventory of $6,000 in the storeroom. During the month, food purchases totaling $8,000 were delivered to the storeroom and food totaling $8,000 was issued...
-
A proton (mass of 1.67 x 10-27 kg) is moving at 1.25 x 106 m/s directly toward a stationary helium nucleus (mass 6.64 x 10-27 kg). After a head-on elastic collision, what is the velocity of the...
-
1. How many degrees Celsius does the temperature decrease when there is a 45.8F decrease in temperature? 2. For each 1.00F change in temperature what is the corresponding change in degrees Celsius?...
-
1. A bowling ball rolls without slipping up a ramp that slopes upward at an angle beta to the horizontal. Treat the ball as a uniform, solid sphere, ignoring the finger holes. What is the...
-
Consider the seventh excited level of the hydrogen atom. A)What is the energy of this level? B)What is the largest magnitude of the orbital angular momentum? C)What is the largest angle between the...
-
A boy on a skateboard coasts along at \(5 \mathrm{~m} / \mathrm{s}\). He has a ball that he can throw at a speed of \(10 \mathrm{~m} / \mathrm{s}\). What is the ball's speed relative to the ground if...
-
"Standard-cost procedures are particularly applicable to process-costing situations." Do you agree? Why?
-
Each reaction is allowed to come to equilibrium, and then the volume is changed as indicated. Predict the effect (shift right, shift left, or no effect) of the indicated volume change. CO(g) + H(g)...
-
(a) The Key Mechanism for Fischer esterification omitted some important resonance forms of the intermediates shown in brackets. Complete the mechanism by drawing all the resonance forms of these two...
-
Most of the Fischer esterification mechanism is identical with the mechanism of acetal formation. The difference is in the final step, where a carbocation loses a proton to give the ester. Write...
-
A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons. (a) Draw the resonance forms of a carboxylic acid that is protonated on the hydroxyl oxygen atom. (b) Compare the...
-
Price, Waterhouse, and Coopers complete their first year of business as a partnership. The partners offer auditing, tax, and advisory services. Use the Tableau Dashboard to determine allocation of...
-
What did economists discover about the American households' retirement saving according to Andrew Biggs' article? Are most American households saving adequately for their retirement, or is there a...
-
Eleanor Inc. creates artisanal crafts for home dcor, the entity has two manufacturing centres: crafting and painting. MOH is allocated based on labour cost in the crafting centre and machine hours in...

Study smarter with the SolutionInn App


