Propose a structure for a compound C15H24O that has the following 1H NMR spectrum. The peak marked
Question:
Propose a structure for a compound C15H24O that has the following 1H NMR spectrum. The peak marked by an asterisk disappears when D2O is added to thesample.
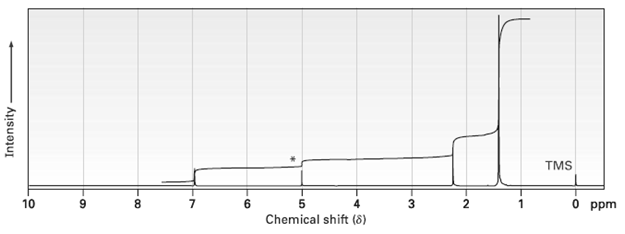
Transcribed Image Text:
TMS 10 8. 0 ppm Chemical shift (8) Intensity 3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
CH33C OH C...View the full answer

Answered By

Sinmon Warui Kamau
After moving up and down looking for a job, a friend introduced me to freelance writing. I started with content writing and later navigated to academic writing. I love writing because apart from making a living out of it, it is also a method of learning and helping others to learn.
5.00+
40+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a structure for a compound with molecular formula C 4 H 8 O that exhibits the following 13 C NMR and FTIR spectra. Carbon NMR 67.7- 25.4- 80 30 20 100 90 70 60 40 10 Chemical shift (ppm) 100...
-
Propose a structure for a compound with molecular formula C 4 H 10 O that exhibits the following 1 H NMR spectrum. Proton NMR 2 3.0 4.0 2.5 Chemical shift (ppm) 2.0 3.5 1.5 1.0
-
Propose a structure for a compound with molecular formula C 10 H 14 O that exhibits the following 1 H NMR spectrum. Proton NMR Chemical shift (ppm)
-
A random queue is a collection that supports the following API: Write a class RandomQueue that implements this API. Use a resizing array. To remove an item, swap one at a random position (indexed 0...
-
Mission statements are often criticized for being lists of platitudes. What can strategic managers do to prevent their statements from appearing to be simple statements of obvious truths?
-
Suppose you are assigned the task of drafting a law to impose a tax on smoke emissions. What provisions would you put into the law? a. How would you decide the size of the tax? b. What would you do...
-
What are the functions in executing and recording these two classes of transactions and custody of related assets?
-
Trieste Toy Company manufactures only one product, Robot Ranger. The company uses a standard cost system and has established the following standards per unit of Robot Ranger: During November, the...
-
Examine the data from the following tables and answer the questions below: U.S. Department of Education, Office of Special Education Programs, Individuals with Disabilities Education Act (IDEA)...
-
Determine the force P required to hold the 50-kg mass in equilibrium. B
-
Compound A, C8H10O, has the IR and 1H NMR spectra shown. Propose a structure consistent with the observed spectra, and assign each peak in the NMR spectrum. Note that the absorption at 5.5 ?...
-
The reduction of carbonyl compounds by reaction with hydride reagents (H: ? ) and the Grignard addition by reaction with organo magnesium halides (R: ? + MgBr) are examples of nucleophilic carbonyl...
-
An FASB member expressed the following view: Are we going to set accounting standards in the private sector or not? . . . Part of the answer depends on how the business community views accounting...
-
Interviewing an elderly with quadriplegic. What did you learn from this experience? How did this interview force you to look at your own life?
-
> For a recent year, McDonald's (MCD) company-owned restaurants had the following sales and expenses (in millions): Sales Food and paper Payroll and employee benefits Occupancy and other expenses...
-
What are the intricate mechanisms underlying the phenomenon of trophic cascades, and how do they manifest within complex ecosystems ?Explain
-
Kampfire, Inc., a very successful manufacturer of camping equipment, is considering "going public" next month to raise funds to help finance the company's future growth. The financial manager of...
-
Monthly loan payment Personal Finance Problem Tim Smith is shopping for a used luxury car. He has found one priced at $35,000. The dealer has told Tim that if he can come up with a down payment of...
-
The dates of meteor showers are rather predictable as listed in Table 28.2. How intense the shower might be, however, is still somewhat of a guess. So keep your eye to the nighttime sky for these...
-
[a] Two foam blocks, each with a charge of 19 micro coulombs (1 C = 10-6 C), are both held in place 19 cm apart in the east-west direction. A foam ball with a charge 49 C is placed 55 cm north of the...
-
A cylinder containing 151 lb Cl 2 has an inside diameter of 10 in. and a height of 45 in. The gas pressure is 100 psi (1 atm = 14.7 psi) at 20 C. Cl 2 melts at -103 C, boils at -35 C, and has its...
-
Heats of hydrogenation of three alkenes are as follows: 2-methyl-1-butene (-119 kJ mol-1) 3-methyl-1-butene (-127 kJ mol-1) 2-methyl-2-butene (-113 kJ mol-1) (a) Write the structure of each alkene...
-
Outline a synthesis of propene from each of the following: (a) Propyl chloride (b) Isopropyl chloride (c) Propyl alcohol (d) Isopropyl alcohol (e) 1, 2-Dibromopropane (f) Propyne
-
Outline a synthesis of cyclopentene from each of the following: (a) Bromocyclopentane (b) Cyclopentanol
-
A recent "study" examined the possibility of cooking a chicken by slapping it really hard. If your hand has enough kinetic energy, the slap could convert this into thermal energy to cook the bird....
-
Skydiving terminal velocity. When determining the terminalvelocity of items in free fall, the friction factor f is typicallycalled the drag coefficient and is represented by the symbol CD.They are...
-
A steel alloy has a melting temperature = 1450C, its density = 7.87 g/cm2, specific heat=0.46 /gC, and heat of fusion - 270 1/8 Assume specific heat has the same value for solid and molten metal. The...

Study smarter with the SolutionInn App


