Propose a mechanism for the followingreaction: HO- CO2CH3 (CH3CH2)3N Heat BRCH2 CO2CH3
Question:
Propose a mechanism for the followingreaction:
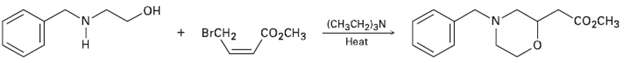
Transcribed Image Text:
HO- CO2CH3 (CH3CH2)3N Heat BRCH2 CO2CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
NH HC proton transfer N Br COCH...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for each reaction. (a) (b) (c) (d) OH H2SO heat OCH + CH3OH H20 CH2OH H2So4 heat CH OCH2CH CH CH,CH2OH (a minor product)
-
Propose a mechanism for the acid-catalyzed condensation of n-propyl alcohol to n-propyl ether, as shown above. When the temperature is allowed to rise too high, propene is formed. Propose a mechanism...
-
Propose a mechanism for the base-promoted hydrolysis of g-butyrolactone:
-
Suppose a single 802.11n client is connected to an 802.11n access point and there is no other client or access point in the neighborhood. The client senses the medium, then sends a 2000-Byte frame,...
-
What information or questions can you use when writing a follow-up message after submitting a résumé?
-
Consider the dissolution of the compound X2Y3, which gives X2Y22+, X2Y4+, X2Y3(aq), and Y2-. Use the mass balance to find an expression for [Y2-] in terms of the other concentrations. Simplify your...
-
Which governmental entities are required to have a single audit?
-
Powell Company began the 2016 accounting period with $40,000 cash, $86,000 inventory, $60,000 common stock and $66,000 retained earnings. During 2016, Powell experienced the following events: 1. Sold...
-
Review the Florida Statute re: Justifiable Use of Force found here: http://www.leg.state.fl.us/statutes/index.cfm?App_mode=Display_Statute&Search_String&URL=0700-0799/0776/Sections/0776.012.html...
-
In Integrative Case 10.1, we projected financial statements for Walmart Stores for Years +1 through +5. The data in Chapter 12, Exhibits 12.17 through 12.19 include the actual amounts for 2015 and...
-
The following transformation involves a conjugate nucleophilic addition reaction (Section 19.13) followed by an intra molecular nucleophilic acyl substitution reaction (Section 21.2). Show...
-
One step in the biosynthesis of morphine is the reaction of dopamine with p-hydroxyphenylacetaldehyde to give S)-norcoclaurine. Assuming that the reaction is acid-catalyzed, propose amechanism. . NH ...
-
How many objects are eligible for garbage collection immediately before the end of the main() method? A. None. B. One. C. Two. D. Three. E. The code does not compile. F. None of the above. public...
-
Crane Leasing Company signs an agreement on January 1, 2025, to lease equipment to Cullumber Company. The following information relates to this agreement. 1. 2. 3. 4. 5. The term of the...
-
On December 1, Jasmin Ernst organized Ernst Consulting. On December 3, the owner contributed $84,780 in assets in exchange for its common stock to launch the business. On December 31, the company's...
-
Solve equation by using the quadratic formula. List the solutions, separated by commas. 4k - k - 4 = 0 k =
-
Kubin Company's relevant range of production is 10,000 to 12,000 units. When it produces and sells 11,000 units, its average costs per unit are as follows: Direct materials Direct labor Variable...
-
An arrow of mass 15 g is shot horizontally into a bale of hay and its strikes the hay with a velocity of 55 m/s. It penetrates a depth of 11 cm before stopping. What is the average stopping force...
-
The following information is available for Chemco Inc.s inventories as of June 30, 2024: We used the LIFO method of valuing our inventories for approximately 40% and 44% of total inventories at June...
-
Nike manufactures shoes and sportswear. How has the Internet changed the way this company communicates with its suppliers and retail customers?
-
Find the pH of a 0.0100 M sulfuric acid (H 2 SO 4 ) solution.
-
Write out the steps in the mechanism for the sulfonation of benzene.
-
Which product would you expect if propene were used in place of ethene in eq. 4.11 (or eqs. 4.20 and 4.21): propylbenzene or isopropylbenzene? Explain. Eq. 4.11 CH2CH3 H2SO alkylation (4.11)
-
Draw the important resonance contributors for the benzenonium intermediate in the bromination of aniline, and explain why ortho, para substitution predominates. NH2 aniline
-
Diaz Company issued $91,000 face value of bonds on January 1, Year 1. The bonds had a 9 percent stated rate of interest and a ten- year term. Interest is paid in cash annually, beginning December 31,...
-
Manvir had to make payments of $1,125 every 6 months to settle a $22,000 loan that he received at 4.52% compounded semi-annually. a. How long did it take to settle the loan?
-
If I invest a single amount of $14,000 in an account earning 8% p.a. compounding quarterly for 5 years, how much interest will I have earned in those 5 years?

Study smarter with the SolutionInn App


