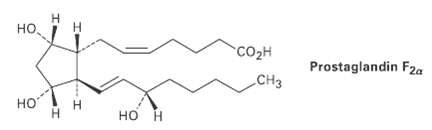
Prostaglandin F2?, a hormone that causes uterine contraction during childbirth, has the following structure. Are the two
Question:
Prostaglandin F2?, a hormone that causes uterine contraction during childbirth, has the following structure. Are the two hydroxyl groups (?OH) on the cyclopentane ring cis or trans to each other? What about the two carbon chains attached to the ring?
Transcribed Image Text:
но, Н н "Cоон Prostaglandin F2a CНз но н Н но н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
H HH COH HO H CH3 Prostaglandin F2 HO H T...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
If a company has the following structure Assets: ........... 10,000 Total Debt: ..........25, 000 Equity Common stock & APIC: .... 5,000 Deficit: .......... (20,000) Total equity: ........ (15,000)...
-
What factors are believed to bring about uterine contractions at the termination of pregnancy?
-
A vicinal diol has OH groups on adjacent carbons. The dehydration of a vicinal diol is accompanied by a rearrangement called the pinacol rearrangement. Propose a mechanism for this reaction. OH OH...
-
Consider a situation with two firms that have marginal abatement cost functions The marginal damage function is again equal to D'(E)=d.E. Assume the regulator applies Monteros mechanism. Determine...
-
Assume that one year ago, you bought 200 shares of a mutual fund for $21 per share, you received an income distribution of $0.11 cents per share, and a capital gain distribution of $0.32 cents per...
-
How do weathering processes affect soil formation?
-
Identify and briefly describe the principal types of (a) processing controls and (b) output controls.
-
Best Buy Co., Inc.s consolidated statements of earnings from its 2009 annual report are presented in Exhibit 5-3.l Required a. Using the statement of earnings, prepare a vertical common-size analysis...
-
8. This is a graph of the function f(x): Give each graph below the correct label from the following: (a) f(x+3) (b) f(x-3) (c) (2x) (d) 2(x) (e)|f(x)| (f) f(x) (g) 2f(x)1 (h) f(2x)+2 (i) f(x) x (j)...
-
Melodic Musical Sales, Inc. is located at 5500 Fourth Avenue, City, and ST 98765. The corporation uses the calendar year and accrual basis for both book and tax purposes. It is engaged in the sale of...
-
Draw the structures of the following molecules: (a) Trans-1-Bromo-3-methylcyclohexane (b) Cis-1, 2-Dirnethylcyclobutane (c) Trails-1-tert-Butyl-2-ethylcyclohexane
-
Names the following substances, including the cis-or trans-prefix (red-brown =Br): (b) (a)
-
Arellano Company uses a periodic inventory system and overstated its ending inventory by $20,000. How will this inventory error affect reported net income for the company?
-
How do you calculate the Yield ANNUAL and Yield - Nominal, Annualized in Excel? Tax rate: 27% Par Value: 1,000 Bond Price; 1,127 Bond Maturity in Years: 20 Bound Coupon Rate: 0.065 Bond Coupon...
-
The fractional concentration of ozone in clean tropospheric air is about 1 x 10-8 at 1 atmosphere pressure and T= 0OC. Calculate this concentration in ppm and also in g/m3.
-
How do you calculate Capital Requirement Ratio when only given an income statement and balance sheet? the formula is (tier 1 capital + tier 2 capital)/risk weighted assets but what makes up tier 1...
-
The following two reactions take place in a gas phase reactor at 3000 K and 1 atmosphere pressure: 2CO 2 (g) 2CO(g) + O 2 (g) K eq .1071 O 2 (g) + N 2 (g) 2NO(g) K...
-
When would it be more advantageous for a firm to use more debt to finance its business? How might this affect the cost and riskiness of the business? How would we determine the cost of both debt and...
-
Your investment portfolio consists of $13,000 invested in only one stockAmazon. Suppose the risk-free rate is 6%, Amazon stock has an expected return of 11% and a volatility of 39%, and the market...
-
Design a circuit which negative the content of any register and store it in the same register.
-
A 25.0 L volume of He(g) at 30.0 C is passed through 6.220 g of liquid aniline (C 6 H 5 NH 2 ) at 30.0 C. The liquid remaining after the experiment weighs 6.108 g. Assume that the He(g) becomes...
-
What is wrong with the use of such names as "isopropanol" and "tert-butanol"?
-
Suggest an experiment using an isotopically labeled alcohol that would prove that the formation of an alkyl sulfonate does not cause cleavage at the C-O bond of the alcohol.
-
An exception to what we have just said has to do with syntheses of unsymmetrical ethers in which one alkyl group is a tert-butyl group and the other group is primary. For example, this synthesis can...
-
SWOT Analysis for Prime Org Strengths . . Powerful brand identity Strong product promotion by celebrities and influencers Robust distribution network Strong customer support Innovation and Product...
-
You are a consultant specializing in L & D, and you have been retained by one of the hotels of Small Ski Resort, a small, traditional ski resort in German speaking Switzerland that has been catering...
-
Imagine that you have been tasked with finding a new CRM software system for your organization. Please research online and identify at least two options you recommend. A few items to include within...

Study smarter with the SolutionInn App


