Provide a structure for each of the following compounds. C 9 H 10 O 3 : IR
Question:
Provide a structure for each of the following compounds.
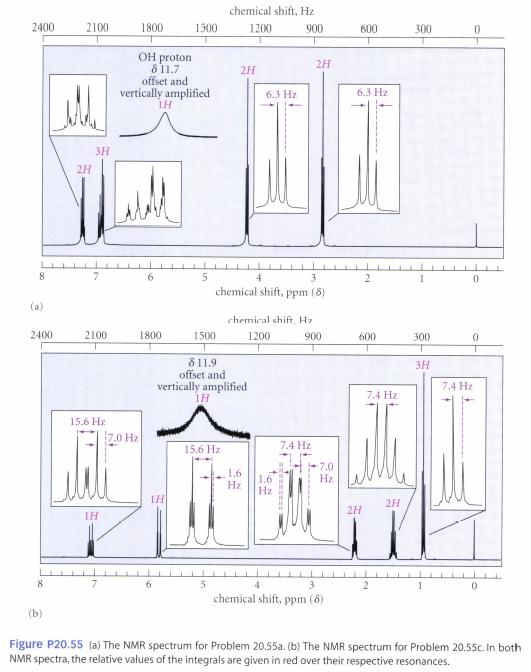
C9H10O3: IR 2400-3200, 1700, 1630 cm–1 NMR: δ 1.53 (3H, t, J = 8 Hz); δ 4.32 (2H, q, J = 8 Hz); δ 7.08, δ 8.13 (4H, pair of leaning doublets, J = l0 Hz); δ 10 (1H, broad, disappears with D2O shake)
Transcribed Image Text:
chemical shift, Hz 2400 2100 1800 1500 1200 600 OH proton 811.7 offset and vertically amplified IH 2H 6.3 Hz 6.3 Hz 3H chemical shift, ppm (8) chemical shift H 2400 2100 1500 δ 11.9 ly amplified 1800 1200 900 offset and 74 H 7.4 Hz 15.6 Hz 7.0 Hz 7.4 Hz 15.6 Hz Hz 2H 21 IH chemical shift, ppm (8) Figure P20.55 (fa) The NMR spectrum for Problem 20.55a. (b) The NMR spectrum for Problem 20.55c. In both NMR spectra, the relative values of the integrals are given in red over their respective resonances.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (18 reviews)
The IR spectrum indicates the presence of a ca...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw a structure for each of the following compounds in its more stable chair conformation. Explain your choice. (a) (b) CH3 CH3 CH (CH),C CHA CH, ," CH3 CH3
-
Give a structure for each of the following compounds. (In some cases, more then one answer is possible) (a) A chiral alcohol C4H6O (b) A diol C4H10O2, that exists in only three stereoiso meric forms
-
Give a structure for each of the following compounds. (a) 3-methyl- 1-octene (b) Isoprene
-
Olmsted Co. has small computer chips assembled in Poland and transports the final assembled products to the parent, where they are sold by the parent in the U.S. The assembled products are invoiced...
-
In October, 2012, seasonally adjusted estimated civilian employment was 143,384,000, while unemployment was 12,258,000 and those out of the labor force numbered 88,341,000. The following month, in...
-
The International Federation of Accountants web address is www.ifac.org. On this page is a section labeled media center. Next, choose articles. Choose one of the items on this page and write a brief...
-
Consider the situation of Example 12.10. Rather than shorting a futures contract, the U.S. firm could borrow \(500 /\left(1+r_{G} ight)\) euros (where \(r_{G}\) is the 90-day interest rate in...
-
The Clearinghouse Sweepstakes has just informed you that you have won $1 million. The amount is to be paid out at the rate of $20,000 a year for the next 50 years. With a discount rate of 10 percent,...
-
Novell, Inc., has the following mutually exclusive projects. Year Project A Project B 0 $22,000 $19,000 1 11,500 12,500 2 8,000 9,000 3 2,800 8,000 a-1. Calculate the payback period for each project.
-
Griffin Companys inventory records for its retail division show the following at December 31: At December 31, 11 of these units are on hand. Journalize the following for Griffin Company under the...
-
Give the structure for an isomer of compound A that has a melting point of 208C and NMR spectra that are almost identical to those of A.
-
Imagine that you have just carried out a conversion of p-bromotoluene into p-bromobenzoic acid and wish to separate the product from the unreacted starting material. Design a separation of these two...
-
In Problem factor completely. If a polynomial cannot be factored, say so. 25m 2 - 16n 2
-
Licking the backs of certain frogs produces visual and hallucinations, memory loss and cognitive dysfunction. What parts of the brain do you think that it affects? What receptors may be involved?
-
how to conduct an external and internal company analysis. Conduct an external analysis of a company or organization of your choosing. Use all of the tools necessary to conduct a thorough evaluation...
-
Why does the sun appear more orange or red when viewed on the horizon, but appears more yellow or cream colored when viewed high in the sky? Explain.
-
Elaborate extensively on the evolutionary trajectories of snakes, crocodiles, lizards, and turtles, elucidating the complex biological adaptations, genetic diversifications, and ecological contexts...
-
What causes surface winds to be weaker than geostrophic winds? Explain.
-
a. Compare the adjustments necessary on the accounts (income or expense only) of Julie Durkin to reflect (1) a cash-based and (2) an accrual-based accounting system (assume the accounting period is...
-
For Problem estimate the change in y for the given change in x. y = f(x), f'(12) = 30, x increases from 12 to 12.2
-
Describe how this amine could be resolved by using this carboxylic acid? CH3 CNH, H HC H CI C COH
-
An unknown compound, X has the formula C6H12. (a) Calculate the degree of un-saturation of X. (b) X reacts with H2 in the presence of a catalyst to form a compound, Y, with the formula C6H14. What...
-
How many stereo isomers exist for this compound? Assign the relative stabilities of each. Is the methyl group axial or equatorial in the more stable conformer of the least stable stereo isomer? CH3 Ph
-
Discuss the main distinguishing features of majority, concurring, and dissenting opinions
-
On September 3, 2023, Wilkins purchased 1244 stock in Brass Corporation for $6,000. On December 31, 2023, the stock was worth $8,500. On August 15, 2024, Wilkins was notified that a. 2023-$0;...
-
Kowalczyk Company spends $30000 dollars for manufacturing equipment. When should the cost of the equipment be expensed? Over the period that the equipment will be used When the performance obligation...

Study smarter with the SolutionInn App


