Rank the following dienophiles in order of their expected reactivity in the Diels?Alder reaction. CH CH NC
Question:
Rank the following dienophiles in order of their expected reactivity in the Diels?Alder reaction.
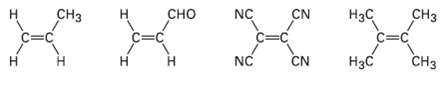
Transcribed Image Text:
CHз CHз NC NC CN Нас c=C CHз н сно c=c Н c=C c=C NC Н CN Н Нас Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (12 reviews)
The most reactive dienophiles contain electronwithdrawing groups Mo...View the full answer

Answered By

Shubhradeep Maity
I am an experienced and talented freelance writer passionate about creating high-quality content. I have over five years of experience working in the field and have collaborated with several renowned companies and clients in the SaaS industry.
At Herman LLC, an online collective of writers, I generated 1,000+ views on my content and created journal content for 100+ clients on finance topics. My efforts led to a 60% increase in customer engagement for finance clients through revamping website pages and email interaction.
Previously, at Gerhold, a data management platform using blockchain, I wrote and published over 50 articles on topics such as Business Finance, Scalability, and Financial Security. I managed four writing projects concurrently and increased the average salary per page from $4 to $7 in three months.
In my previous role at Bernier, I created content for 40+ clients within the finance industry, increasing sales by up to 40%.
I am an accomplished writer with a track record of delivering high-quality content on time and within budget. I am dedicated to helping my clients achieve their goals and providing exceptional results.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Rank the following dienophiles (from least reactive to most reactive) in terms of reactivity in a Diels-Alder reaction:
-
Rank the following substances in order of their expected SN1 reactivity: Br Br H CHCCH CHCH2Br H2C=CHBr
-
Rank the following halides in order of their reactivity in the Williamson synthesis: (a) Bromo ethane, 2-bromopropane, bromo benzene (b) Chloro ethane, bromo ethane, 1-iodopropene
-
The Wall Street Journal Corporate Perceptions Study 2011 surveyed readers and asked how each rated the quality of management and the reputation of the company for over 250 worldwide corporations....
-
What are some of the benefits of offering a non-traditional benefits package to employees such as the one Google offers?
-
Obtain the magnitude plot of the response V0 in the network of Fig. 14.106 for the frequency interval 100 Figure 14.106 50 10 AF
-
For a mixture of \(70 \mathrm{~mol} \%\) chloroform, \(15 \mathrm{~mol} \%\) acetone, and \(15 \mathrm{~mol} \%\) ethanol at \(1 \mathrm{~atm}\), show on a residue curve map the feasible compositions...
-
Prior to instituting an activity-based costing system, Harris and Whelan, the attorneys in E9-5 and E9-6, utilized a simplified costing system with one direct cost category, professional labor, and...
-
Suppose you have just purchased a 10-year General Electric 100 million zero-coupon bond at 8% YTM. The exchange rate is USD1.1/EUR. What will be your rate of return if one year from now the YTM on...
-
The following T accounts show transactions that were recorded in December 20X1 by Cedar Canyon Nursery and Landscape, a firm that specializes in residential and commercial landscaping. The entries...
-
Reaction of isoprene (2-methyl-1, 3-hutadiene) with ethyl propenoate gives a mixture of two Diels?Alder adducts. Show the structure of each, and explain why a mixture is formed. CO2CH2CH3
-
1, 3-Cyclopentadiene is very reactive in DielsAlder cyclo addition reactions, but 1, 3-cyclohexadiene is less reactive and 1, 3-cycloheptadiene is nearly inert. Explain. (Molecular models are...
-
On what balance sheet accounts does the issuance of common stock have an effect?
-
What are the best ratio to determine the credit worthiness of a company? Explain all steps.
-
Using the data below: a) calculate the variance B) calculate the composition of the minimum variance portfolio comprised of securities A and B. C) calculate the variance of minimum variance portfolio...
-
In March of 2008, theM/V MSC Sabrinaran aground in the St. Lawrence River near Trois-Rivires, Quebec, Canada. Tugboats made initial attempts to free the vessel, but they were ultimately unsuccessful...
-
1. Given 2 equations of the IS curve, Y = 2500-800r and Y = 2500 -1500r: a) Calculate the national income equilibrium (Y) for both equations if r = 5% and 10%. b) Based on the IS equations, draw the...
-
A compay reported 31, 2024, balance sheet equipment at cost of $800,000.a disclosure note indicates that the company uses straight line depreciation over five years, and estimates salvage value as...
-
Southeast Shoe Distributor (SSD) is a closely owned business that was founded 10 years ago by Stewart Green and Paul Williams. SSD is a distributor that purchases and sells mens, womens, and...
-
Identify one local business that uses a perpetual inventory system and another that uses a periodic system. Interview an individual in each organization who is familiar with the inventory system and...
-
What is the vapor pressure of a solution in which the mole fraction of the solute is 0.200 and the vapor pressure of the pure solvent is 100.0 torr? (Assume a single nonvolatile, nonelectrolyte...
-
Rank the following compounds in order of increasing amount of hydrate present at equilibrium. Br Br CHO CHO
-
NMR spectra for two compounds are given here, together with the molecular formulas. Each compound is a ketone or an aldehyde. In each case, show what characteristics of the spectrum imply the...
-
Show how you would accomplish the following syntheses. (a) Cyanohydrins acetorphenone cyanohydrin (b) Cyclopentanecarbaldehyde 2-cyclopentyl-2-hydroxyacetic acid (c) hexan-1-ol 2-hydroxyheptanoic...
-
The board chair has asked management to develop some strategies to improve profitability and estimate the impact of the strategies on the hospital's return on equity (ROE). By how much would the 2021...
-
Discuss the ethical consderariion relayed to the use of information rechnology for organizational control how can organization ensure responsibility and transparent?
-
Can we justify the acquisition of Peloton Interactive Inc using NPV assuming the initial investment is 10% of the total market value of the company, 5% constant growth rate for the next three years...

Study smarter with the SolutionInn App


