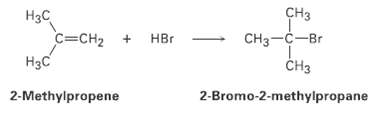
Reaction of HBr with 2-methyipropenc yields 2-hromo-2-methylpropane, what is the structure of the carbocation formed during the
Question:
Reaction of HBr with 2-methyipropenc yields 2-hromo-2-methylpropane, what is the structure of the carbocation formed during the reaction? Show the mechanism of the reaction.
Transcribed Image Text:
CHз CHз—с—Br Нас C=CH2 + HBr Нас сHз 2-Bromo-2-methylpropane 2-Methylpropene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
The mechanism is pictured in Figure The steps 1 Attack of the electrons of th...View the full answer

Answered By

Sumit kumar
Education details:
QUATERNARY Pursuing M.Tech.(2017-2019) in Electronics and Communication Engg. (VLSI DESIGN) from
GNIOT Greater Noida
TERTIARY B.Tech. (2012-2016) in Electronics and Communication Engg. from GLBITM Greater Noida
SECONDARY Senior Secondary School Examination (Class XII) in 2012 from R.S.S.Inter College, Noida
ELEMENTARY Secondary School Examination (Class X) in 2010 from New R.J.C. Public School ,Noida
CERTIFICATION
Summer Training in ‘WIRELESS EMBEDDED SYSTEM’ from ‘XIONEE’ for the six weeks.
EMBEDDED SYSTEM Certificate issued by CETPA INFOTECH for one day workshop.
Certificate of Faculty development program on OPTICAL COMMUNICATION and NETWORKS for one week.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Reaction of HBr with 3-methylcyclohexene yields a mixture of four products: cis- and trans-1-bromo-3-methylcyclohexane and cis- and trans-1-bromo- 2-methylcyclohexane. The analogous reaction of HBr...
-
Reaction of HBr with (R)-3-methyl-3-hexanol leads to racemic 3-brorno-3-mcthylhcxane.Explain. OH CH3CH2CH2CCH2CH3 3-Methyl-3-hexanol H3
-
What is the structure of ATCase?
-
Suppose that in a particular area the consumption of water varies tremendously throughout the year, with average household summer use exceeding winter use by a great deal. What effect would this have...
-
Assume you are 59 years old, want to retire in 6 years, and currently have an investment portfolio valued at $550,000 invested in technology stocks. After talking with friends and relatives, you have...
-
Suppose you want to test whether girls who attend a girls' high school do better in math than girls who attend coed schools. You have a random sample of senior high school girls from a state in the...
-
A face-centered cubic array of round fibers is shown in Figure 3.6. Derive the relationship between the fiber volume fraction and the given geometrical parameters. What is the maximum possible fiber...
-
Surreal Sound, Inc., manufactures and sells compact disks. Price and cost data are as follows: Selling price per unit (package of two CDs).................. $ 25.00 Variable costs per unit: Direct...
-
A Treasury bond with maturity exactly 18 years and 5 months and coupon 5.0 percent trades at an yield to maturity (YTM) of 3.3 percent (BEY). What is the value of the bond (and, therefore, its dirty...
-
Figure shows a uniform beam subject to a linearly increasing distributed load. The equation for the resulting elastic curve is w. (-X* + 21X - L*X) 120 EIL Based on the above equation determine the...
-
What product would you expect from reaction of cyclohexene with HBr? With HC1 HBr
-
Add curved arrows to the following polar reactions to indicate the flow of electrons in each: :Ci: (a) - :- + -N :CI: -NH (b) H CH: -Br: :Br: (c) :0: :CI: CI "
-
Find the perimeter of the nth figure in Exercise 34. Exercise 34.
-
When does the certify MA/DA statement button become enabled so you can click to certify the managing account?
-
In this lesson, you have learned about the concept of "post-truth." Use the attached worksheet to introduce yourself to me and to demonstrate your understanding of the notion of post-truth and...
-
When customer service workers start in their new positions, what type of support do they receive?
-
When reviewing an applicant's driving history, how many years do insurance companies request information for with respect to the applicant's former convictions and license suspensions/cancellations?
-
Name the levels in Maslows hierarchy and give an example of a marketing appeal that is focused at each level.
-
The following selected information was taken from Sun Valley Citys general fund statement of revenues, expenditures, and changes in fund balance for the year ended December 31, 2019: Revenues:...
-
In which of the following is the central atom sp hybridized? (a) BeCl 2 ; (b) BCl 3 ; (c) CCl 4 ; (d) NCl 3 ; (e) None of these.
-
A compound (N) with the molecular formula C9H10O reacts with osmium tetroxide. The 1H NMR spectrum of N is shown in Fig. 14.31 and the IR spectrum of N is shown in Fig. 14.32. Propose a structure for...
-
The IR and 1H NMR spectra for compound X (C8H10) are given in Fig. 14.33. Propose a structure for compound X.
-
The IR and 1H NMR spectra of compound Y (C9H12O) are given in Fig. 14.34. Propose a structure for Y.
-
Should an MNC purchase Nestl stock yes or no? Recommendations should include the company's sustainability plans, the image of the company, Gordon Growth result, and financial performance. Any...
-
Can the sponsor, or general partner, continue to achieve such high returns? Framed another way, can the general partner earn a 25+% return on the $6,305,000 received at the end of the 10th year? If...
-
Select a hospital that publicly displays their financial performance on their website. Describe the Profit Loss Statement: Review top revenue items Review cost comparison compared to previous year...

Study smarter with the SolutionInn App


