Refer to the footnote in Table 14-1. How many seconds will it take for (a) H +
Question:
Refer to the footnote in Table 14-1. How many seconds will it take for (a) H+ and (b) NO-3 to migrate a distance of 12.0 cm in a field of 7.80 × 103 V/m?
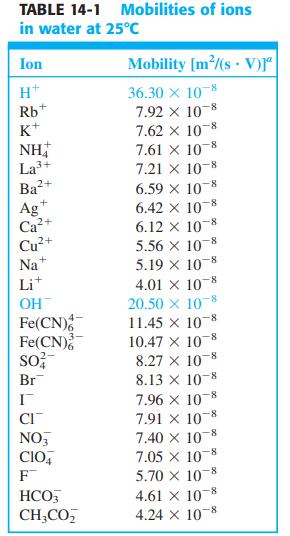
Transcribed Image Text:
TABLE 14-1 Mobilities of ions in water at 25°c Ion Mobility [m/(s V)r" 36.30 x 108 7.92 x 10-8 7.62 x 10 7.61 X 10 8 7.21 x 10-8 H* Rb K+ NH La3+ Ba2+ 6.59 X 10 6.42 x 10-8 6.12 x 10 5.56 X 10-8 +, Ag Ca²+ Cu+ Na* 8- 5.19 x 10 4.01 x 10-8 20.50 x 10 11.45 x 10-8 10.47 X 10-8 8.27 x 10 8.13 x 10-8 Lit OH Fe(CN) Fe(CN) so; Br 7.96 X 10 7.91 x 10 7.40 X 108 7.05 x 10 5.70 x 10-8 CI NO, CIO, F 4.61 X 10-8 - 4.24 X 10-8 HCO, CH;CO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Velocity mobility field 3630 10 8 m 2 s V 780...View the full answer

Answered By

Pranav Makode
I am a bachelor students studying at professor ram meghe institute of technology and research. I have a great experience of being an expert. I have worked as an expert at helloexperts and solvelancer as a part time job. I have also worked as a doubt solver at ICAD SCHOOL OF LEARNING, which is in Amravati city. I have also worked as an Freelancer.
I have great experience of helping students, as described above. I can help any students in a most simple and understandable way. I will not give you have any chance for complaint. You will be greatfull to accept me as an expert.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
1. How many seconds would it take a 7.00-hp motor to raise a 475-lb boiler to a platform 38.0 ft high? 2. How long would it take a 950-W motor to raise a 360-kg mass to a height of 16.0 m?
-
How many seconds are there in a solar year (365.24 days)?
-
How many time constants will it take for a charged capacitor to be discharged to one-fourth of its initial stored energy?
-
Write a method leve1Order() that prints BST keys in level order: first print the root; then the nodes one level below the root, left to right; then the nodes two levels below the root (left to...
-
Define a provision and give an example.
-
What is the definition of ignorable treatment assignment? a. Give an example of a study where the treatment assignment is ignorable. b. Give an example of a study where the treatment assignment is...
-
Derive the equation of motion for free transverse vibration of a simply supported, specially orthotropic plate that is subjected to in-plane loads per unit length \(N_{x}\) and \(N_{y}\) as shown in...
-
Oxmoor Manufacturing Co. expects to make 40,000 chairs during the 2013 accounting period. The company made 4,000 chairs in January. Materials and labor costs for January were $20,000 and $30,000,...
-
How are best fit and best practice HR practices similar and different, what are the benefits and limitations of each?
-
On January 1, 2020 High Ltd. Purchased 70% of the shares of Low Ltd. for $2,100,000 and on the same day, Low Ltd. purchased 65% of the shares of Middle Ltd. for $1,950,000. Any fair value...
-
Which side of the liquid junction 0.1 M KNO3 | 0.1 M NaCl will be negative?
-
Suppose that an ideal hypothetical cell such as that in Figure 13-7 were set up to measure E for the half-reaction . (a) Calculate the equilibrium constant for the net cell reaction. (b) If there...
-
Answer each of the following questions related to various short-term liabilities: a. On September 1, 2016, a company borrowed $100,000 from its bank and signed a nine-month note with 8% interest. The...
-
What is the effect on the equilibrium price and equilibrium quantity of theater tickets if the price of an orchestra ticket decreases and the wage rate paid to actors decreases? The equilibrium price...
-
Based on readings provided on this module, critically evaluate a recent Big Tech competition case and ruling at an EU level that you are familiar with, with a focus on ( i ) the market failure issue,...
-
Use Diagrams A and B to complete the following sentence: The charge separation distance in B is times greater than in A; the force of attraction is A B 14 in B. A. 1st Blank: eight; 2nd Blank: four...
-
Consider two isolated, charged conducting spheres: a large sphere and a second smaller sphere with a radius 10 times smaller than that of the large sphere, but with 6 times as much charge. (a)...
-
A wrecking ball rotates in a vertically circular path with a radius of 12.9 m when it strikes a building. Determine the maximum velocity it can strike the building at safely if the cable can...
-
Suppose that the random sample in problem 8 yielded these observed scores: a. Find the 95% confidence interval for the mean. b. Find the 99% confidence interval for the mean. 6 5 6 12 5 10 11 13 12...
-
Use translations to graph f. f(x) = x-/2 +1
-
A 3.455-g sample of a mixture was analyzed for barium ion by adding a small excess of sulfuric acid to an aqueous solution of the sample. The resultant reaction produced a precipitate of barium...
-
A tanker truck carrying 5.0 103 kg of concentrated sulfuric acid solution tips over and spills its load. If the sulfuric acid is 95.0% H2SO4 by mass and has a density of 1.84 g/mL, how many...
-
A sample of 5.53 g of Mg(OH)2 is added to 25.0 mL of 0.200 M HNO3. (a) Write the chemical equation for the reaction that occurs. (b) Which is the limiting reactant in the reaction? (c) How many moles...
-
Vodaphone's earnings per share in 2022 was $1.80, and in 2017 it was $1.25. The company distributes 60% of its earnings as dividends, and the stock is currently valued at $37.75. The cost to issue...
-
A bond is currently selling in the market for $1,085.96. It has a coupon of 8% and a 15-year maturity. Using annual compounding, what is the yield to maturity on this bond?
-
Korda International Inc. recently issued new securities (common shares and bonds) to finance a new project with a cost $16 million. The equity issued had a flotation cost of 8%, while the debt issued...

Study smarter with the SolutionInn App


