Referring to Exercise 21-C, predict the relative amounts of C 2 H 2 79 Br 2 ,
Question:
Referring to Exercise 21-C, predict the relative amounts of C2H2 79Br2, C2H2 79Br81Br, and C2H2 81Br2 in 1,2-dibromoethylene. Compare your answer with Figure 21-7.
Figure 21-7
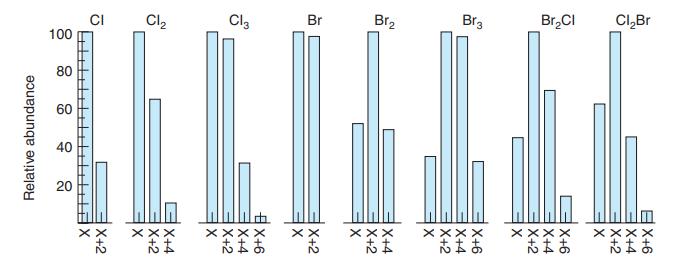
Transcribed Image Text:
CI 100 Cl2 Cl, Br Br2 Br3 Br,CI CI,Br 80 60 40 20 XX く× もまあ XXXX もまあ Relative abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
79 Br abundance 0506 9 8lBrabundance b 04931 Abundanc...View the full answer

Answered By

Ranjan kumar
I Graduated from IIEST Shibpur in 2017 and Currently I am Working in Microsoft for the past 1.7 years. I am a very fun loving and friendly guy who loves to impart knowledge in his free time. I am also associated with Online Tutoring for the past 1 year and have helped more than 1000 Students till now . I gel up with students pretty quickly and train them in the required subjects with utmost care.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
From the natural abundance of 79 Br and 81 Br, predict the relative amounts of CH 79 Br 3 , CH 79 Br 2 81 Br, CH 79 Br 81 Br 2 , and CH 81 Br 3 . As in Exercise 21-C, the fraction of each isotopic...
-
Referring to Exercise 21-C, predict the relative abundances of 10B2H6, 10B11BH6, and 11B2H6 for diborane (B2H6).
-
The temperature-composition diagram for the Ca/Si binary system is shown in Fig. 6.46. (a) Identify eutectics, congruent melting compounds, and incongruent melting compounds. (b) If a 20 per cent by...
-
In the context of supply and demand under international trade, when will a country decide to export a particular good? Import a good? Who gains and loses under each decision?
-
R. Santiago Co. uses special journals and a general journal. The following transactions occurred during May 2014. May 1 R. Santiago invested $40,000 cash in the business. 2 Sold merchandise to Lawrie...
-
Brice Foran Hair Care has a balance date of 30/6/X1. You have reviewed their accounts and found that several adjustments need to be made prior to the preparation of the final financial statements....
-
True or False. Dry friction can introduce nonlinearity in the system.
-
Denny Manufacturing had a bad year in 2012. For the first time in its history, it operated at a loss. The companys income statement showed the following results from selling 80,000 units of product:...
-
https://www.cbc.ca/player/play/831149123630 Who is Helen Knott? How does that make the topic of this talk so significant?
-
Show that 1 minus the Jaccard similarity is a distance measure between two data objects, x and y, that satisfies the metric axioms given on page 70. Specifically, d(x, y) = 1 J(x, y).
-
Briefly describe how a magnetic sector mass spectrometer works.
-
Find the number of rings plus double bonds in molecules with the following compositions and draw one plausible structure for each: (a) C 11 H 18 N 2 O 3 ; (b) C 12 H 15 BrNPOS; (c) Fragment in a mass...
-
An ideal gas expands while the pressure is kept constant. During this process, does heat flow into the gas or out of the gas? Justify your answer.
-
Evaluate the integral J 2x +3 x + x dx
-
In n + 1 space dimensions we have Cartesian coordinates a = (1, n+1) and spherical polar coordinates (r. 2), where :-) (...) are n angles. They are related by ra]. The integral over all space can be...
-
As part of the program to reduce smoking, the Ministry of Health ran an advertising campaign to convince people to quit smoking. To evaluate the effectiveness of their campaign, they had 12 subjects...
-
Project the future revenue growth rate for the next three years of the company. Explain the assumptions you used to predict the company's revenue growth rate(s) for the next three years. Project the...
-
For each of the following transactions that occur in their lives, identify whether it is included in the calculation of U.S. GDP as part of consumption (C), investment (1), government purchases (G),...
-
Match the following normalized impedances with points A, B, C, D, and E on the Smith chart of Figure 11.44. (i) 0 + j0 (ii) 1+ j0 (iii) 0 j1 (iv) 0 + j1 (v) + j (vi) [Zin / Z]min (vii) [Zin / Z]max...
-
Listed below are common types of current liabilities, contingencies, and commitments: a. Accounts payable b. Bank loans and commercial paper c. Notes payable d. Dividends payable e. Sales and excise...
-
(a) Write the Nernst equations for the half-reactions in Demonstration 14-1. In which direction do electrons move through the circuit? (b) If you use your fingers as a salt bridge in Demonstration...
-
Write a balanced chemical equation (in acidic solution) for the reaction represented by the question mark.22 As in Box 14-5, calculate Eo for the reaction. 1.441 1.491 1.584 1.098 BrO,- HOB - -...
-
What must be the relation between E o+ and E o- if the species X+ is to diproportionate spontaneously under standard conditions to X 3+ and X(s)? Write a balanced equation for the disproportionation.
-
Compute ending work in process inventory for a manufacturer using the following information. Raw materials purchased Direct materials used Direct labor used Factory overhead Work in process...
-
A spring with spring constant 80 N/m is placed at the bottom of a 6 m high ramp with opening angle 60 . A 3 kg mass is then placed on the spring and pushed down so the spring is compressed by a...
-
A 2kg mass is moving at 10m/s along a frictionless surface towards a spring (at rest) with spring constant 50N/m. When the mass hits the spring, it will begin to compress it. What is the maximum...

Study smarter with the SolutionInn App


