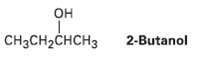
(S)-2-Butanol slowly racemizes on standing in dilute sulfuric acid.Explain. CH3CH2CHCH3 2-Butanol...
Question:
(S)-2-Butanol slowly racemizes on standing in dilute sulfuric acid.Explain.
Transcribed Image Text:
Он CH3CH2CHCH3 2-Butanol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (17 reviews)
Since 2butanol is a secondary alcohol substitution can occur by e...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
On standing in dilute aqueous acid, compound A is smoothly converted to mevalonolactone. Suggest a reasonable mechanism for this reaction. What other organic product is also formed? CH3 CH3 CHj ...
-
On standing in dilute aqueous acid, compound A is smoothly converted to mevalonolactone.
-
(R)-2-Pentanol racemizes when placed in dilute sulfuric acid. Draw a mechanism that explains this stereochemical outcome, and draw an energy diagram of the process.
-
The probability that fewer than 35 people support theprivatization of Social Security A discrete random variable is given. Assume the probability of the random variable will be approximated using the...
-
What is knowledge management and why is it so important to today's employers?
-
Show how you might use a nucleophilic substitution reaction of 1-bromopropane to synthesize each of the following compounds. (You may use any other compounds that are necessary.) (a) (b)...
-
You have audited the financial statements of the Heft Company, a December 31 year end client, for several years. The interim phase of the engagement, completed on August 31, included confirming...
-
A new systems development project is Pete's first experience as a project manager, and he has led his team successfully to the programming phase of the project. The project has not always gone...
-
An experimental rocket sled accelerates from 12.0 m/s to 280 m/s in 4.70 seconds. How far did it travel in this time?
-
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that...
-
Reaction of HBr with (R)-3-methyl-3-hexanol leads to racemic 3-brorno-3-mcthylhcxane.Explain. OH CH3CH2CH2CCH2CH3 3-Methyl-3-hexanol H3
-
Using Kurt Lewins change model, give a force field analysis for Holdens challenges in delivering innovative products. Suggest how the company might overcome resistance and foster permanent change.
-
4. Beach Bums is a popular beachside store. Becky, owner of the Beach Bums, is deciding how much refrigerator space to devote to four different drinks. Pertinent data on these four drinks are as...
-
On August 31, 2024, the general ledger of Brusher Man Vacuum shows a balance for cash of $7,904. Cash receipts yet to be deposited into the checking account total $3,336. The company's balance of...
-
If a 200 keV photon hits an electron at the K-edge and it is absorbed by the photoelectric effect, what is the relativistic momentum and velocity of the electron?
-
What causes an object to move? What is the role of the net force or sum of forces that is greater than zero acting on an object? Compare the motion (or acceleration) of an object (of mass 2.0 kg)...
-
Cooperative San Jos of southern Sonora state in Mexico makes a unique syrup using cane sugar and local herbs. The syrup is sold in small bottles and prized as a flavoring for drinks and desserts. The...
-
Two events in reference frame \(\mathrm{S}\) occur \(10 \mu \mathrm{s}\) apart at the same point in space. Frame \(\mathrm{S}^{\prime}\) travels at speed \(v=0.90 c\) relative to frame...
-
Coastal Refining Company operates a refinery with a distillation capacity of 12,000 barrels per day. As a new member of Coastal's management team, you have been given the task of developing a...
-
Write an expression for the equilibrium constant of each chemical equation. a. SbCl, (g) SbCl3(g) + Cl(g) b. 2 BINO (g) = 2NO(g) + Br(g) c. CH(g) + 2 HS(g) = CS(g) + 4 H(g) d. 2 CO(g) + O(g) = 2 CO(g)
-
Predict the products from crossed Claisen condensation of the following pairs of esters. Indicate which combinations are poor choices for crossed Claisen condensations. (a) (b) (c) (d) Ph -CH2 C-OCH,...
-
Show how crossed Claisen condensations could be used to prepare the following esters. (a) (b) (c) (d) Ph C-CH-C OCH,CH CH CH-C-OCH3 C-C-oCH Eto C CH-C OCH,CH Ph CH,CH,CH,
-
Predict the major products of the following crossed Claisen condensations. (a) (b) (c) NaOCH + Ph-C OCH NaOCH,CH CH,CH C CH CH,CHO C 0CH2CH NaOCH2CH
-
The wheels of a skateboard roll without slipping as it accelerates at 0.45 m/s down an 80-m-long hill. Part A If the skateboarder travels at 1.5 m/s at the top of the hill, what is the average...
-
A 2.30 g air-inflated balloon is given an excess negative charge, q = -3.50 10-8 C, by rubbing it with a blanket. It is found that a charged rod can be held above the balloon at a distance of d =...
-
A positive charge q = 2.60 C on a frictionless horizontal surface is attached to a spring of force constant k as in the figure shown below. When a charge of 42 = -8.350 C is placed 9.50 cm away from...

Study smarter with the SolutionInn App


