Show how ethyl bromide can be used as a starting material in the preparation of each of
Question:
Show how ethyl bromide can be used as a starting material in the preparation of each of the following compounds.
(a)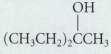
(b)
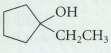
Transcribed Image Text:
он (CH CH2) CCH он СН,СН,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
In each case ethyl bromide CH 3 CH 2 Br react...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In the previous section, we learned how to use malonic ester as a starting material in the preparation of substituted carboxylic acids (the malonic ester synthesis). That method employed a step in...
-
Decamethonium bromide is used in surgery as a muscle relaxant. It acts by preventing the enzyme acetylcholine esterase from destroying acetylcholine (see page 327), a necessary step in the...
-
Phosgene (CC1 2 O) is a colorless gas that was used as an agent of chemical warfare in World War I. It has the odor of newly mown hay (which is a good warning if you know the smell of newly mown...
-
Upton Computers makes bulk purchases of small computers, stocks them in conveniently located warehouses, ships them to its chain of retail stores, and has a staff to advise customers and help them...
-
Benson and Orton are partners who share income in the ratio of 1:3 and have capital balances of $70,000 and $30,000 respectively. Ramsey is admitted to the partnership and is given a 40% interest by...
-
Match each of the key terms with the definition that best fits it. ________ Detailed information about a systems design specifications, its internal workings, and its functionality. Here are the key...
-
The pipe lies in the horizontal plane. If it is subjected to a vertical force \(\mathbf{P}\) at its end, determine the strain energy due to bending and torsion. Express the results in terms of the...
-
Provence Bicycle Company manufactures road bikes. The following data for September of the current year are available: Quantity of direct labor used ...... 650 hrs. Actual rate for direct labor...
-
what are your opinion regarding Evaluating Database Security and Cyber Attacks paper? here's the link:...
-
Refer to the Big Rig Rental Company case. Design a spreadsheet that will allow the firm to determine the Net Present Value of cash flows over the five-year period. The following exercises refer to...
-
Within set, which compound should be more reactive in carbonyl-addition reactions? Explain your choices. H,CCC CH or HCC CH2CH --
-
Outline two different Grignard syntheses for 3-methyl-2-butanol.
-
President Donald Trump spent much of his time in office erecting barriers to trade with other nations, but in early 2020 his administration signaled that it wanted to do something different:...
-
Halo Corporation, a U.S. corporation, has a foreign office that conducts business in Portugal. Halo pays foreign taxes of $55,000 on foreign-source taxable income of $189,750. Its U.S.-source taxable...
-
Ross' employer withheld $ 1 , 9 2 0 federal income tax and $ 9 0 0 New York income tax from his paycheck in 2 0 2 3 . Ross also has a $ 2 5 0 New York income tax overpayment from 2 0 2 2 tax year. He...
-
Ali and Yousef established AY partnership on January 1, 2020. Each one invested 20000. On March 1, All invest 2000, on June 1 Yousef invests 4000. On September 1, Ali withdraws 1000. On November 1,...
-
A refrigerator has a power rating of 225W. It runs for 24 hours per day for a full year (365 days). a. How many hours does it run in one year? b. How much energy does it use in 1 year in kWh? ...
-
Use intercepts to graph each equation 6x-2y-12 = 0
-
Based on the U.S. Census population projections for 2016 to 2060, the projected Asian population (in millions) can be modeled by the exponential function A(t) = 14.8311.01592t, where t = 0...
-
For the following exercises, write the polynomial function that models the given situation. Consider the same rectangle of the preceding problem. Squares of 2x by 2x units are cut out of each corner....
-
The energy level diagram for the MOs of CH 2 = CH 2 is shown if Figure 3.24. Show a similar diagram for the lowest-energy excited state of this molecule. C=C 5 o* * Ethene 5 o Energy
-
Consider the species formed by the addition of an extra electron to H2 so that there are three electrons and a negative charge. Show an energy level diagram for the MOs of this species. Is there...
-
Draw an energy level diagram for the excited state of H2. Is there still a bond between the hydrogens?
-
Ida Company produces a handcrafted musical Instrument called a gamelan that Is similar to a xylophone. The gamelans are sold for $949. Selected data for the company's operations last year follow:...
-
There is a bonus with the following characteristics: nominal value $98.650.000 annual coupon rate 4.5% term 5 years, tir 6.5% A) Calculate the price of the bond assuming it is zero coupon B)...
-
A project that costs $4,450 to install will provide annual cash flows of $1,500 for each of the next 6 years. a. What is NPV if the discount rate is 11%? b. How high can the discount rate be before...

Study smarter with the SolutionInn App


