Show how you could prepare the following compounds from 4-methyl-3-penten-2-one, (CH3)2C =CHCOCH3. (b) (c) CHCCHCH2C (a)
Question:
Show how you could prepare the following compounds from 4-methyl-3-penten-2-one, (CH3)2C =CHCOCH3.
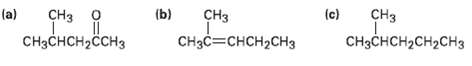
Transcribed Image Text:
(b) (c) сНз CHзC—CHCH2CНЗ (a) CHз CHз CHзCнсH2сH2сHз CHзснсH2ссHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
CH3 CH3CCHCCH3 a H P...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show how you would prepare the following compounds from the appropriate sulfonyl chlorides. (a) (b) (c) SO,CH oSO2Me
-
Show how you could prepare the following amino acidenantioselectively:
-
Show how you could prepare the following amino acids using a reductive amination: (a) Methionine (b) Isoleucine
-
Imagine that you are a human resource manager in a domestically based company that has decided to operate internationally. You have been charged with sorting out the HRM effects of the decision. What...
-
Use IRS publications and other reference materials to answer a specific tax question. Contact an IRS office to obtain an answer for the same question. What differences, if any, exist between the...
-
Use synthetic division of find the quotient and the remainder. a. (x3 + 2x2 - 13x + 10) ( (x - 5) b. (x4 + 3x3 + 3x2 + 3x + 2), ( (x + 2) c. (x5 - 2x) ( (x + 1)
-
What are COTS products and NDIs and how do they relate to items and CIs?
-
Presented below is information related to Garland Co. for the month of January 2014. Instructions (a) Prepare the necessary adjusting entry for inventory. (b) Prepare the necessary closingentries....
-
While driving her BMW X5 2020 model, with registration number AD 54 XX GP, along R556 in Johannesburg, Ms Enne Martins was crashed into by a gas tanker truck belonging to EC Fuels (Pty) Ltd. The...
-
1. Why did Sainsbury bid for Argos? 2. With reference to the post-acquisition integration matrix (see Figure 11.3) consider how Sainsbury might best integrate Argos? 3. With reference to the buy,...
-
Draw the following molecule as a line-bond structure, and show how it can be prepared from a ketone and an amine.
-
Show all the steps in the acid-catalyzed formation of a cyclic acetal from ethylene glycol and an aldehyde or ketone.
-
Lukow Products is investigating the purchase of a piece of automated equipment that will save $400,000 each year in direct labor and inventory carrying costs. This equipment costs $2,500,000 and is...
-
Explain what is meant by corporate governance. In September 2014, Alibaba listed on the New York Stock Exchange in what was, at the time, the largest IPO in history. Explain some of the reasons that...
-
How does the standard define relevance and reliability? What must an auditor do if conditions indicate that a document may not be authentic or the terms in a document have been modified but that the...
-
Assume that CEDE Insurance Company has purchased from SS Reinsurance Company a surplus share treaty with a $5,000,000 limit and a retention of $70,000. CEDE has written three policies. Policy A...
-
Ariel files his tax return as married filing jointly ( MFJ ) . He runs a general store as a sole proprietor with no employees. In 2 0 2 3 , his qualified business income ( QBI ) of $ 2 0 0 , 0 0 0...
-
Assume that ABC Insurance Company has purchased from QS Reinsurance Company a quota share treaty with a $500,000 limit and a retention of 30 percent and a cession of 70 percent. ABC has written...
-
Emmanuel Adewale, 25 years old, works in Abuja as a customer relationship manager at a major bank. His current personal portfolio is made mostly of bonds (60%), value stocks (20%), and ETFs (20%)...
-
The Ranch 888 Noodle Company sells two types of dried noodles:ramen, at $6.50 per box, and chow fun, at $7.70 per box. So farthis year, the company has sold a total of 110,096 boxes ofnoodles,...
-
Formaldehyde gas, H 2 CO, is used in the manufacture of plastics; in aqueous solution, it is the familiar biological preservative called formalin. Describe the molecular geometry and a bonding scheme...
-
Write structural formulas for the major organic products from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) O (excess), H SO (cat.) OH 0 H2O, H,so, (cat.) (1) Mg (2) CO2 (3) H2O Br...
-
Indicate reagents that would accomplish each of following transformations. More than one reaction may be necessary in some cases. (a) (b) (c) (d) (e) (f) OCH HO CI Cl OCH3 NH OH ENH,CI
-
Which acid of each pair shown here would you expect to be stronger? (a) (b) (c) (d) (e) HO HO 10 HO HO 3 10 F O or OH OH or MeJN OH OH or CF
-
Describe a time when you had to stop a client's violent or destructive behaviour. What did you do? What would you have done differently?
-
Analyze the integration of digital technologies, such as process simulation, computational fluid dynamics (CFD), and advanced control systems, in achieving process intensification, focusing on...
-
When monochromatic light of an unknown wavelength falls on a sample of copper, a minimum potential of 2.49 V is required to stop all of the ejected photoelectrons. (The work function for copper is...

Study smarter with the SolutionInn App


