Show the molecular ions formed from these compounds: b) a) CH,NHCH,
Question:
Show the molecular ions formed from these compounds:
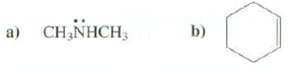
Transcribed Image Text:
b) a) CH,NHCH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
a C...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Compounds A and B are isomeric amines of molecular formula C8H11N. Identify each isomer on the basis of the 1H NMR spectra given in Figure 22.9. Question continue over next page Figure 22.9 Compound...
-
Compounds A and B are isomers of molecular formula C9H19Br. Both yield the same alkene C as the exclusive product of elimination on being treated with potassium tert-butoxide in dimethyl sulfoxide....
-
Compounds A and B are isomers of molecular formula C9H19Br. Both yield the same alkene C as the exclusive product of elimination on being treated with potassium tert-butoxide in dimethyl sulfoxide....
-
?
-
Identify the major stakeholders in your organization, discuss there different types.
-
What reactive, learning, behavioral, and results/ outcome measures do you propose to assess the effects of this training program? Write at least one specific example of each type of evaluative...
-
As you are standing on the platform, a boy in a train moving to the right extends his hand outside the window and releases a ball. In what direction is the ball moving according to (a) you (b) the...
-
Dan Murphy awoke at 5:45 A.M., just like he did every workday morning. No matter that he went to sleep only four hours ago. The Orange Bowl game had gone late into the evening, and the New Years Day...
-
7. A vertical spring is fixed to one of its end and a massless plank fitted to the other end. A block is released from a height has shown. Spring is in relaxed position. Then choose the correct...
-
A ride hailing company has their DB structured in 3 major tables as described in the SCHEMA section below. Write a query to fetch the top 100 users who traveled the most distance using the service....
-
What conclusions can be drawn about these compounds from their massspectra? 100 43 55 135 85 69 107 164 trpt 40 50 60 70 80 L10 120 130 90 100 140 150 160 170 m/z b) 100 73 50 20 30 40 50 60 70 80 90...
-
Show equations for the major fragmentations you would expect from the molecular ions of these compounds. List the m/z of the productions. CH3 CH2 a) CH;CH-CH,CH-CH; b) CH;CH,CHCH2CH3
-
On January 2, 20X0, Kowalski Construction Company signed a contract to provide paved roads to a new housing development. The project will last 2 years, and the total payment will be $4million, to be...
-
A 71.0-mH inductor and a 7.30-F capacitor are connected in series with a generator whose frequency is 232 Hz. The rms voltage across the capacitor is 1.80 V. Determine the rms voltage across the...
-
Use object-oriented design to design a class to manage a share portfolio. Shareholdings are identified by the company name, the number of shares purchased, the date of the purchase, the cost per...
-
A project with expected NOI of $500 000 next year. NOI grow at 3% thereafter, forever. If the required rate of return 13 % and a new building cost is $ 4 000 000, what is the value of land? 2. A...
-
Chevy Corp. has common shares with a current total market value of $75,000,000, and a cost of equity of 29%. The company has total debt outstanding that has a market value of $60,000,000 and a cost...
-
On November 1st, 2020, Gracie Cosmetics took out a loan of 209,842 to build a new location by the town mall. The bank charges them an annual rate of 9% beginning on November 1st. Gracie Cosmetics...
-
Which of the following is the correct accounting treatment to record the provision for onerous contracts? a. Debit Provision for Onerous Contracts (Contra-Asset), Credit Provision for Onerous...
-
Sundial Technologies produces and sells customized network systems in New Brunswick. The company offers a 60-day, all software and labor-and an extra 90-day, parts-only- warranty on all of its...
-
The World Bank was established in 1944. To date, it has lent nearly $390 billion, mostly to poor nations. However, its accomplishments have been questioned. For example, since 1951, India has...
-
Write bond-line formulas for three esters with the formula C5H10O2.
-
Write another resonance structure for ethyl acetate. Include formal charges.
-
Write another resonance structure for acetamide.
-
Shaw Company has the following account balances: Receivables $ 1 0 0 , 0 0 0 Inventory 1 5 0 , 0 0 0 Land 1 0 0 , 0 0 0 Building net 2 5 0 , 0 0 0 Liabilities 1 0 0 , 0 0 0 Common stock 1 0 0 , 0 0 0...
-
Mizan Corporation, a manufacturing company, prepares its financial statements annually. During the last month of the fiscal year, Mizan experiences a significant surge in sales. The sales team closes...
-
A firm uses job - order costing. Current period facts: Actual overhead: $ 1 0 4 , 6 8 8 Applied overhead: $ 1 0 4 , 9 0 5 Budgeted overhead: $ 1 0 8 , 6 9 8 COGS balance ( before closing ) : $ 3 9 0...

Study smarter with the SolutionInn App


