Show the products of thesereactions: 1) Hg(O,CCH,) . 2) NaBH3. NAOH 1) Hg(O,CCH,)2, H,0 2) NaBH4, NaOH
Question:
Show the products of thesereactions:
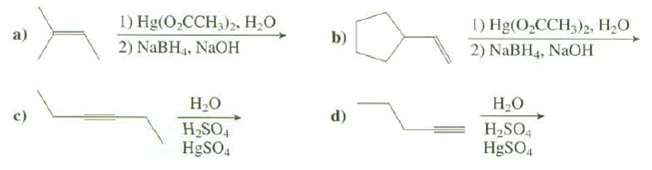
Transcribed Image Text:
1) Hg(O,CCH,)» Н.О 2) NaBH3. NAOH 1) Hg(O,CCH,)2, H,0 2) NaBH4, NaOH b) a) Н.о H SO4 Н.о H2SO4 d) HgSO, H9SO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (12 reviews)
a...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
Calculate the density of N2 at STP, (a) using the ideal-gas law and (b) using the molar volume and molar mass of N2. How do the densities compare?
-
Wilson revises her estimates of the benefits from the new system's lower labor costs as calculated in P1-28A. She now thinks the savings will be only $935,000. Requirements 1. Compute the expected...
-
A rigid, vertical rod of negligible mass is connected to the floor by a bolt through its lower end, as shown in FIGURE 11-45. The rod also has a wire connected between its top end and the floor. If a...
-
A marketed asset's price \(x\) is governed by the mean reverting process where \(\eta, \theta\), and \(\sigma\) are positive constants and \(z\) is a Wiener process. (a) Let \(V(x, t)\) be a given...
-
The Lubbock plant of Morrils Small Motor Division produces a major subassembly for a 6.0 horsepower motor for lawn mowers. The plant uses a standard costing system for production costing and control....
-
Compute the integral (x.ex)dx. (x-e3-x)dx dx Click here to start next step
-
One piece of PVC pipe is to be inserted inside another piece. The length of the first piece is normally distributed with mean value 20 in. and standard deviation .5 in. The length of the second piece...
-
Show all the steps in the mechanism for the formation of MTBE from methanol and isobutylene.
-
Explain which of the reaction would provide a better synthesis of3-hexanone. . . CH,CH,CH,CH,CH, 3-Hexanone CH,CH,C=CCH CH3 CH,C=CCH,CH,CH3 H,SO4 HgSO, H&SO,
-
Write the SQL code to change the EMP_PCT value to 3.85 for the person whose employee number (EMP_NUM) is 103. Next, write the SQL command sequences to change the EMP_PCT values as shown in...
-
A 70.0-kg person throws a 0.0500-kg snowball forward with a ground speed of 33.5 m/s. A second person, with a mass of 59.5 kg, catches the snowball. Both people are on skates. The first person is...
-
One barn, a unit used for cross sections in nuclear and particle physics, is equal to 1 0 ^ 2 8 1 0 - 2 8 square meters, about the cross - sectional area of a uranium nucleus. Assuming an atom is...
-
Gayle runs at a speed of 4.45 m/s and dives on a sled, initially at rest on the top of a frictionless snow-covered hill. After she has descended a vertical distance of 5.00 m, her brother, who is...
-
A 0.034-kg bullet is fired vertically at 208 m/s into a 0.15-kg baseball that is initially at rest. How high does the combined bullet and baseball rise after the collision, assuming the bullet embeds...
-
A1,170-kgcar traveling initially with a speed of25.0 m/sin an easterly direction crashes into the rear end of a9,200-kgtruck moving in the same direction at20.0 m/s(see figure below). The velocity of...
-
Talk to your parents about their taxes. Do they follow any of the five tax reduction strategies presented in this chapter? Talk to them about the benefit of following one or more of these strategies....
-
4. Jobe dy -Y 2 et by
-
Calculate the value of the equilibrium constant for each of the following reactions in aqueous solution. a. HCHO + OHCHO + HO b. C,H,O, +H*
-
Predict the product(s) of the following reactions. If more than one product is formed, tell which ismajor. (a) CH3I (excess) Ag20, H20 eat C? A? B? .cocI (b) H20 NaN3 eat C? B? A? (c) CgHsCH2Br C7...
-
Fill in the missing reagents a?c in the following scheme: NH2 CHCH3 CH=CH2 CCH3 b, c CHCH2NCH3 CH CH-CH2
-
Although pyrrole is a much weaker base than most other amines, it is a much stronger acid (pK a 15 for the pyrrole versus 35 for diethyl amine). The NH proton is readily abstracted by base to yield...
-
Popular furniture company, IKEA, has purchased forests in Romania as well as land in Alabama to assist with keeping up with the wood demand necessary to complete customer orders. This was one way...
-
How does China being Turkey's biggest import partner affect Turkey's exchange rate?
-
Assignment 4 In this assignment you are provided information on an experiment and you are required to investigate and interpret the output which is provided below. Problem: Consider the...

Study smarter with the SolutionInn App


