Show the rearranged carbocations that are expected from these carbocations: a) +CH CH3 b) CHCHCHCHCH c) CH3
Question:
Show the rearranged carbocations that are expected from these carbocations:
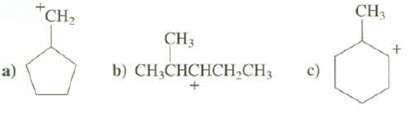
Transcribed Image Text:
a) +CH₂ CH3 b) CH₂CHCHCH₂CH₂ c) CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
A carbocation may rearrange to form a more stable ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show that a conditionally convergent series can be rearranged so to diverge?
-
Carbocations are stabilized by hyperconjugation: Tertiary are the most stable, followed by secndary. Primary and methyl cations are too unstable to form in solution.
-
The acid-catalyzed dehydration of an alcohol to a rearranged alkene is known as a Wagner-Meerwein rearrangement. Propose a mechanism for the following Wagner-Meerwein rearrangement: OH ...wr + H30+...
-
1. What is Ladures target market and retail strategy in the United States? 2. Explain the reasons Ladure owns its stores in some countries and uses franchising with local licensees in others. 3....
-
Orbital Industries Ltd manufactures a variety of materials and equipment for the aerospace industry. A team of P & D engineers in the firm's Technology Park plant has developed a new material that...
-
On January 1, year 1, LaGuardia Company signed a five-year non-cancellable lease for a new machine with a fair value of $ 80,000, requiring $ 8,000 annual payments at the beginning of each year. The...
-
How do you write a Problem Statement?
-
ANALYSIS OF PROFITABILITY Based on the financial statement data in Exercise 24-1A, compute the following profitability measures for 20-2 (round all calculations to two decimal places): (a) profit...
-
Write two complex numbers z = a + bi and z2 = c + d with a, b, c and d not equal to 0 or 1 with a product of 4+221.
-
1. Assess the extent of the accommodations the employer made to allow Ciha to be able to return to work. 2. Because Bradley Ciha suffered no loss of earnings, how can he be considered 80 percent...
-
Show both the substitution and elimination products that are formed in these reactions: a) C CI + CH0 CHOH + OH HO EtOH b) Br + CHOH CH3OH
-
Show the substitution products for these reactions: a) b) +CHCHOH Br CI Ja + HO EtOH HO EtOH
-
Find the derivative using the appropriate rule or combination of rules. y = (kx + b) 1/3 ; k and b any constants
-
(b) Given the distribution of 8 bits output complied Table Q2(b). Determine the conditions or values of the C, Z, DC, N and OV flags for each of the following: Table Q2(b) Bit 7 X (i) (ii) Bit 6 X...
-
The following questions involve showing that double encryption using various ciphersystems is not better than a single encryption. In every case, be specific, that is, explain how the single...
-
Using the grammar rules given below, write down the derivation of the following statements: Grammar: - Your answer > = , => (+1-) | , => | (* | /) , => A|B|C|D A = A + B C
-
(Consider the encryption function for a symmetric cryptosystem described by the table below, where K = M = C = {0, 1, 2, 3} k 0 m 1 03 0 2 1 1 3 0 22 1 0 30 2 3 2 3 1 2 3 1 (i) Give the corresponding...
-
Assume that an administrator was checking a switch configuration, he/she typed the command show vlan brief and got the following output: VLAN Name 1 default 20 MKTG 30 SALES 40 R&D 50 PROD 1002...
-
Uber is a service people use to arrange transportation with drivers who use their own cars for this purpose. Customers pay for their rides with the Uber smartphone app. Ubers prices fluctuate with...
-
A container holds 2.0 mol of gas. The total average kinetic energy of the gas molecules in the container is equal to the kinetic energy of an 8.0 10-3-kg bullet with a speed of 770 m/s. What is the...
-
Solve each inequality and graph its solution. -3b + 5 + 7 < 14-b -9-8-7-654-3 -2 -1 0 1
-
Draw and name all monochloro products you would expect to obtain from radical chlorination of 2-methylpentane. Which, if any, are chiral?
-
Taking the relative reactivities of 1 o , 2 o , and 3 o hydrogen atoms into account, what product(s) would you expect to obtain from monochlorination of 2-methylbutane? What would the approximate...
-
Draw three resonance forms for the cyclohexadienylradical. Cyclohexadienyl radical
-
entory LO P1 Compute ending work in process inventory for a manufacturer using the following information. Raw materials purchased Direct materials used Direct labor used Factory overhead Work in...
-
Four departments of a company need to order the following amounts of the same products. (a) Use matrix multiplication to get a matrix showing the comparative costs for each department for the...
-
The trial balance columns of the worksheet for Cullumber Company at June 30, 2027, are as follows. Cullumber Company Worksheet For the Month Ended June 30, 2027 Trial Balance Account Titles Dr. Cr....

Study smarter with the SolutionInn App


