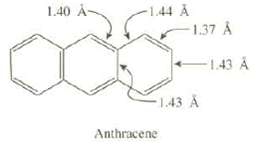
Show the three additional resonance structures for anthracene. Discuss whether the experimental bond lengths shown in the
Question:
Show the three additional resonance structures for anthracene. Discuss whether the experimental bond lengths shown in the following structure are in accord with predictions based on these resonance structures:
Transcribed Image Text:
1.40 A- -1.44 A -1.37 A -1.43 A Anthracene -1.43 A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
The longest bonds 144 and 143 are single in 3 structures ...View the full answer

Answered By

Rodrigo Louie Rey
I started tutoring in college and have been doing it for about eight years now. I enjoy it because I love to help others learn and expand their understanding of the world. I thoroughly enjoy the "ah-ha" moments that my students have. Interests I enjoy hiking, kayaking, and spending time with my family and friends. Ideal Study Location I prefer to tutor in a quiet place so that my students can focus on what they are learning.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The resonance structures of carbon monoxide are shown below. Show how each structure can be converted into the other using the curved-arrow notation. :C-: :C=0:
-
For each of the structures below, draw the resonance structure that is indicated by the curved arrows. Be sure to include formal charges. a. b. c. d. e. f. g. h. N-
-
The following two drawings are resonance structures of one compound: But the following two drawings are not resonance structures: They are, in fact, two different compounds. Explain. Not resonance...
-
Cost-Volume-Profit (CVP) analysis can be used to determine the effect of changes in costs and volumes on a company's net profits. This project assignment assumes that you are newly hired as an...
-
Clarke Auto Parts Company manufactures replacement parts for car repairs. The company recently installed a flexible manufacturing system (FMS), which has significantly changed the production process...
-
Prior to liquidating their partnership, Ellis and Ericson had capital accounts of $17,000 and $29,000, respectively. Prior to liquidation, the partnership had no cash assets other than what was...
-
Water flows in a rectangular channel at a rate of \(q=20 \mathrm{cfs} / \mathrm{ft}\). When a Pitot tube is placed in the stream, water in the tube rises to a level of \(4.5 \mathrm{ft}\) above the...
-
A balance sheet contains the following classifications: (a) Current assets (b) Investments (c) Property, plant, and equipment (d) Intangible assets (e) Other noncurrent assets (f) Current liabilities...
-
Watch the video of the Frontline documentary about questionable police shootings by the New Orleans Police Department in the wake of Hurricane Katrina. Develop an essay that discusses the possible...
-
Assuming the existence of roots, show that if c > 1, then c1/m < c1/n if and only if m > n.
-
Phenanthrene has five total resonance structures. One is shown here. Draw the other four. Which carbon-carbon bond of Phenanthrene would your predict to be the shortest? Phenanthrene
-
One general reaction of radicals is the coupling of one with another to form a bond, as shown in the following equation: The following coupling reaction gives two products. Show the structures of...
-
Which of the following accounts would not appear in the post-closing trial balance? Interest Payable; Equipment; Depreciation Expense; Jennifer Shaeffer, Drawing; Unearned Revenue; Accumulated...
-
Driving down a long straight highway, you see a reflection of the sky in the road ahead. It disappears before you get to it but reappears down the road continually. Because there is nothing different...
-
What is the payback period for the project described in Problem 4.21 if the maintenance increases by $\$ 500 /$ year (after the first year)? Data from in problem 4.21 What is the payback period for...
-
Why is dynamic modeling based on objects, but not classes?
-
A light ray travels up from the bottom of a tank containing mineral water (index of refraction \(n=1.37\) ). If the water surface is smooth, at what minimum angle from the normal to that surface...
-
A healthcare home-care firm has a profit margin of 15 percent on visiting nurse sales of $\$ 20,000,000$. If the firm has debt of $\$ 7,500,000$, total assets of $\$ 22,500,000$, and an after-tax...
-
\(\sigma_{1}
-
On January 2, 20X3, Sheldon Bass, a professional engineer, moved from Calgary to Edmonton to commence employment with Acco Ltd., a large public corporation. Because of his new employment contract,...
-
(a) Approximate the function f(x) = 3 x by a Taylor polynomial of degree 2 at a = 8. (b) How accurate is this approximation when 7 x 9?
-
Hydrocortisone, a naturally occurring hormone produced in the adrenal glands, is often used to treat inflammation, severe allergies, and numerous other conditions. Is the indicated ?OH group in the...
-
A1, 2-cis disubstituted cyclohexane, such as cis-1, 2-dichlorocyclohexane, must have one group axial and one group equatorial. Explain.
-
A 1, 2-trans disubstituted cyclohexane must have either both groups axial or both groups equatorial. Explain.
-
Why are gains/losses added back to (or deducted from) the cash from operations section of a cash flow statement?
-
Combination A, B, C, and F is the fifth most valuable with an NPV of $169,000. how did you came up with it?
-
Using a suitable example explain how you would check that equipment is ready for shut down to commence?

Study smarter with the SolutionInn App


