A small bead with a mass of 100 g slides along a semicircular wire with a radius
Question:
A small bead with a mass of 100 g slides along a semicircular wire with a radius of 10 cm that rotates about a vertical axis at a rate of 2 revolutions per second, as in Figure. Find the values of ? for which the bead will remain stationary relative to the rotating wire.
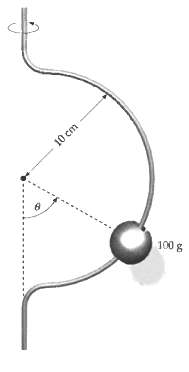
Transcribed Image Text:
10 cm 100 g
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
The semicircular wire of radius 10 cm limits the motion of the bead in the sa...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Mechanics questions
-
A smooth horizontal disc rotates about the vertical axis O (Fig. 4.15) with a constant angular velocity (o. A thin uniform rod AB of length l performs small oscillations about the vertical axis A...
-
A carnival merry-go-round rotates about a vertical axis at a constant rate. A man standing on the edge has a constant speed of 3.66 m/s and a centripetal acceleration d of magnitude 1.83 m/s2....
-
A ball of positive charge rotates about a vertical axis (Fig. P20.61). (a) If the rotation is clockwise as viewed from above, what is the direction of the balls magnetic moment? (b) If the charge on...
-
1. Calculate the density of O2, in g/L, at STP. 1.62 g/L b. 0,714 g/L 1.43 g/L. d. 1.14 g/L e. 0.810 g/L a. .
-
How does the magnitude of the electrical force between a pair of charged particles change when the particles are moved half as far apart? One-third as far apart?
-
What is the conservative position on national health insurance? What is the liberal position? What is your view toward national health insurance? Justify your answer. Visit the Web site of the...
-
\(\left(x^{6} ight)^{2}\) Simplify the expression.
-
Equity Securities Entries Capriati Corporation made the following cash purchases of securities during 2010, which is the first year in which Capriati invested in securities. 1. On January 15,...
-
1. List at least three main differences between the audit report of non-public entities and that of public entities. 2. Provide an example of a situation where the "design of controls" may be...
-
The trial balance of Pacilio Security Services Inc. as of January 1, 2020, had the following normal balances: Cash ..................$122,475 Petty cash ................ 100 Accounts receivable...
-
An object on the equator has an acceleration toward the center of the earth because of the earth's rotation and an acceleration toward the sun because of the earth's motion along its orbit. Calculate...
-
Consider a bead of mass m that is free to move on a thin, circular wire of radius r. The bead is given an initial speed v 0 , and there is a coefficient of kinetic friction k. The experiment is...
-
Complete the following table, indicating the sign and amount of the effect ( 1 for increase, 2 for decrease, and NE for no effect) of each transaction. Provide an account name for any revenue or...
-
Name eight examples of risk that management should identify and control.
-
When auditing individual accounts and disclosures, an auditor's objective is to determine the validity of management's a. Calculations. b. Recording process. c. Assertions. d. Internal control.
-
Differentiate between a self-locking and self-energizing brake.
-
Why do auditors make preliminary estimates of materiality for the individual accounts in the financial statements?
-
A population has mean = 47.5 and standard deviation = 12.6. A sample of size 112 is drawn. a. Find the probability that x is between 45 and 48. b. Would it be unusual for x to be greater than 48?...
-
The quantity Q3 Q1 is known as the ________________. In Exercises 912, fill in each blank with the appropriate word or phrase.
-
The figure shows six containers, each of which is filled from the top. Assume that water is poured into the containers at a constant rate and each container is filled in 10 seconds. Assume also that...
-
The [CrCl 6 ] 3 ion has a maximum in its absorption spectrum at 735 nm. Calculate the crystal field splitting energy (in kJ/mol) for this ion.
-
A model airplane with mass 0.750 kg is tethered by a wire so that it flies in a circle 30.0 m in radius. The airplane engine provides a net thrust of 0.800 N perpendiculars to the tethering wire. (a)...
-
The combination of an applied force and a friction force produces a constant total torque of 36.0 N,m on a wheel rotating about a fixed axis. The applied force acts for 6.00 s. During this time the...
-
A firefighter, a distance d from a burning building, directs a stream of water from a fire hose at angle i above the horizontal as in Figure P4.20. If the initial speed of the stream is vi, at what...
-
Read the scenario located in "Problems - Series A," section 8-19A of Ch. 8, "Performance Evaluation," of Fundamentals of Managerial Accounting Concepts . This scenario puts you in charge of pr...
-
F of 2 Book Hint Required information Use the following information for the Exercises 3-7 below. (Algo) [The following information applies to the questions displayed below] Laker Company reported the...
-
Anita Strods opened a business called Strods Engineering and recorded the following transactions in its first month of operations. June 1 Anita Strods, the owner, invested $156,000 cash, office...

Study smarter with the SolutionInn App


