Some radio-nuclides decay by capturing one of their own atomic electrons, K- shell electrons, say. An example
Question:
Some radio-nuclides decay by capturing one of their own atomic electrons, K- shell electrons, say. An example is below. Show that the disintegration energy Q for this process is given by, where mV and mTi, arte the atomic masses of 49V and 49Ti, respectively, and EK is the binding energy of the vanadium K-shell electron.
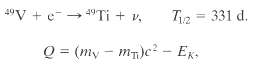
Transcribed Image Text:
4°V + e-- 49TI + v. = 331 d. T2 Q = (my – mT)c? - Ex.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Assuming the neutrino has negligible mass then Amc mmymc Now since Vanadium has 23 electrons see A...View the full answer

Answered By

Subash Murugaih
I am leading expert in this web site couple of years and My clients are much happy with my works and services.
4.60+
309+ Reviews
539+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Modern Physics questions
-
Show that the disintegration energy for decay is Q = (mp mD me)c2 = (Mp MD)c2, where the ms represent the masses of the parent and daughter nuclei and the Ms represent the masses of the neutral...
-
Show that the disintegration energy for decay is Q = (mp mD me)c2 = (Mp MD 2me)c2, where the ms represent the masses of the parent and daughter nuclei and the Ms represent the masses of the...
-
Find the disintegration energy Q for the decay of 49V by K-electron capture. The needed data are mv = 48.948 52u, mTi = 48.947 87 u, and Ek = 5.47keV.
-
Its estimated that the average corporate user sends and receives some 112 e-mails daily.78 Thats about 14 e-mails per hour, and even if half of those dont require a lot of time and concentration,...
-
Wilson Marketing Consulting, a proprietorship owned by Lin Wilson, was started on January 1, 2013, with an investment of $50,000 cash. The company prepares marketing plans for clients. It has been...
-
Which property of the third-row nonmetallic elements might be the one depicted in the graph on the next page: (a) First ionization energy (b) Atomic radius (c) Electronegativity (d) Melting point (e)...
-
A rectangular cartop carrier of \(1.6-\mathrm{ft}\) height, \(4.0-\mathrm{ft}\) length (front to back), and 4.2-ft width is attached to the top of a car. Estimate the additional power required to...
-
After the partnership has been operating for a year, the Capital accounts of Bob and Kim are $15,000 and $10,000, respectively. The firm has cash of $12,000 and office equipment of $13,000. The...
-
Losing to a Weaker Foe What began as a heavily conventional military campaign to unseat the regime of Saddam Hussein had become a bitter, unconventional struggle against frustrated Sunnis who...
-
1. What should Theresa put in the marketing segment? What types of information will she need? 2. For the critical risks assessment segment, what key areas does Theresa have to address? Discuss two of...
-
The cesium isotope 137Cs is present in the fallout from aboveground detonations of nuclear bombs. Because it decays with a slow (30.2y) half-life into 137Ba, releasing considerable energy in the...
-
The radionuclide 11 C decays according to 11 C 11 B + e + + v, T 1/2 = 20.3. The maximum energy of the positions is 0.960MeV. (a) Show that the designation energy Q for this process is given by Q =...
-
What is the functional type of mannitol salt agar (MSA)? Explain how it is utilized in the isolation and identification of Staphylococcus species.
-
1. Formulate a research topic on an issue of interest to you. Refer to your notes on how to formulate a problem and come up with an interesting and researchable topic. (10 marks) 2. Identify the...
-
discuss the emerging paradigm of circadian regulation in coordinating metabolic rhythms across diverse tissues and how disruptions in circadian homeostasis contribute to the pathogenesis of metabolic...
-
Considering a MARR of 12%, Assembly A Assembly B Assembly C First cost, $ ($175,000) ($160,000) ($145,000) Annual cost, $/year ($25,000) ($19,000) ($12,000) Salvage Value, $ $9,000 $10,000 $12,000...
-
The Winterland DPW can purchase road salt from three different mines. Salt from any of these mines must be ordered in 100 ton quantities. Nearby DPW directors that Sarah spoke with stated lead times...
-
Based on key consumer rates such as the Prime and Federal Funds rates, would you describe these rates as: neutral, low or high at this time. (answer in at least a paragraph)
-
A fund manager has a well-diversified portfolio that mirrors the performance of an index and is worth $360 million. The value of the index is 1,200, and the portfolio manager would like to buy...
-
Without solving, determine the character of the solutions of each equation in the complex number system. 3x 2 3x + 4 = 0
-
A certain DAC has the following specifications: eight-bit resolution, full scale = 2.55 V, offset 2 mV; accuracy = 0.1% F.S. A static test on this DAC produces the results shown in Table 11-11. What...
-
Two long straight wires with equal cross-sectional radii a are located parallel to each other in air. The distance between their axes equals b. Find the mutual capacitance of the wires per unit...
-
A long straight wire is located parallel to an infinite conducting plate. The wire cross-sectional radius is equal to a, the distance between the axis of the wire and the plane equals b. Find the...
-
Find the capacitance of a system of two identical metal balls of radius a if the distance between their centers is equal to b, with b >> a. The system is located in a uniform dielectric with...
-
What are some scholarly sources that support the implementation of a case manager?
-
Review your own current processes relating to ER and IR within the organisation and identify three issues that can be improved upon.
-
A group of students working on a collaborative research project needs a centralized space to store and edit their documents. Which Teams feature should they primarily use?

Study smarter with the SolutionInn App


