Spectroscopic data for the indicators thymol blue (TB), semithymol blue (STB), and methylthymol blue (MTB) are shown
Question:
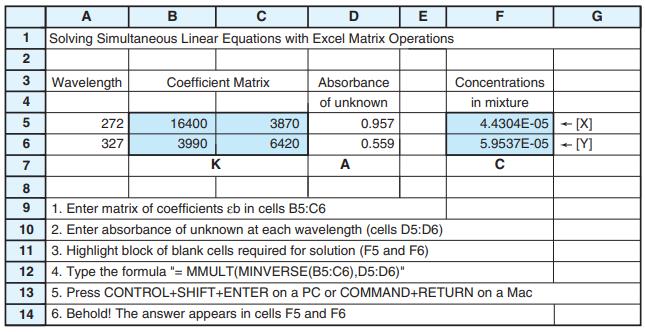
Spectroscopic data for the indicators thymol blue (TB), semithymol blue (STB), and methylthymol blue (MTB) are shown in the table. A solution of TB, STB, and MTB in a 1.000-cm cuvet had absorbances of 0.412 at 455 nm, 0.350 at 485 nm, and 0.632 at 545 nm. Modify the spreadsheet in Figure 18-5 to handle three simultaneous equations and find [TB], [STB], and [MTB] in the mixture.
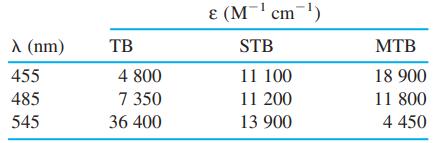
Figure 18-5
Transcribed Image Text:
E (M' cm-) A (nm) TB STB MTB 455 4 800 11 100 18 900 485 7 350 11 200 11 800 545 36 400 13 900 4 450
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (8 reviews)
1 23456789 A B D E F Solving Simultaneous Linear Equat...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Figure 13.44 presents several types of spectroscopic data (IR, 1H NMR, 13C NMR, and mass spectra) for a particular compound. What is it? Figure 13.44 e 100 80 E 60 2 40 86 0 22 0 10 20 30 40 50 60 70...
-
What spectroscopic method could you use to distinguish among the following three isomeric acids? Tell what characteristic features you would expect for eachacid. CH3(CH213CO2H (CH/2CHCH-cO2H...
-
UV-Vis, IR, NMR, and mass spectrometry are spectroscopic tools we use to obtain structural information about compounds. For each pair of compounds below, describe at least one aspect from each of two...
-
Suppose you wanted to hasten the transition from a depletable fossil fuel to solar energy. Compare the effects of a per-unit tax on the depletable resource to an equivalent per-unit subsidy on solar...
-
Rigney Inc. uses the allowance method to estimate uncollectible accounts receivable. The company produced the following aging of the accounts receivable at year-end. Instructions (a) Calculate the...
-
When a suspected drunk driver blows 188 mL of his breath through the fuel-cell breathalyzer described in Section 20.7, the breathalyzer produces an average of 324 mA of current for 10 s. Assuming a...
-
True or False: The damage shown in the image below, due to EDM, takes several years to occur.
-
Why is organizational diagnosis essential to the success of any change effort?
-
For the following compound identify how many chiral carbons present and how many isomers are possible. (6 points) Br HO HO Figure out elements of unsaturation for the following compound. C5H9OBr 1....
-
Iris Kroneman approaches you with the following information (a) She resides in a province with a 5% corporate tax rate (i.e., a total tax rate of 16%) where she owns and operates an incorporated...
-
Infrared spectra are customarily recorded on a transmittance scale so that weak and strong bands can be displayed on the same scale. The region near 2 000 cm -1 in the infrared spectra of compounds A...
-
The spreadsheet gives the product b for four pure compounds and a mixture at infrared wavelengths. Modify Figure 18-5 to solve four equations and find the concentration of each compound. You can...
-
Refer to the facts in the preceding problem. a. If the business is operated as a partnership, explain the payroll tax/self-employment tax implications for the entity, Thomas, and Angela. (No...
-
Refer to Table 16-6. If the Bank of Springfield has lent out all the money it can given its level of deposits, then what is the reserve requirement?
-
Yusuf wants to buy a machine but he currently does not have enough fund, hence he needs to save money for that purpose. If he manages to save RM420 per month and earn annual rate of 6% compounded...
-
Austin bought an annual perpetuity due in 2 0 0 2 that pays $ 4 on "even" years ( i . e . 2 0 0 2 , 2 0 0 4 , 2 0 0 6 , etc. ) and $ 7 on "odd" years ( i . e . 2 0 0 3 , 2 0 0 5 , etc. ) . Calculate...
-
how effective is strain theory in explaining crime?
-
How does the demographic transition in the United States affect public health practices at all levels?
-
After you have studied Economics in the News on pp. 46 47, answer the following questions. a. How does the discovery of new oil and gas reserves change Canadas PPF ? b. How do technological advances...
-
Consider a closed, rigid tank with a volume of 0.8L, filled with cold water initially at 27C. The tank is filled such that there are no voids (air pockets) within. The initial pressure within the...
-
Why is it not practical to titrate an acid or a base that is too weak or too dilute?
-
Calculate the pH of a solution made by mixing 50.00 mL of 0.100 M NaCN with (a) 4.20 mL of 0.438 M HClO 4 (b) 11.82 mL of 0.438 M HClO 4 (c) What is the pH at the equivalence point with 0.438 M HClO...
-
Calculate the pH when 25.0 mL of 0.020 0 M 2-aminophenol have been titrated with 10.9 mL of 0.015 0 M HClO 4 .
-
Ramirez Company installs a computerized manufacturing machine in its factory at the beginning of the year at a cost of $45,300. The machine's useful life is estimated at 10 years, or 403,000 units of...
-
14. (3 points) Write a program that ask the user for 1. their first name and 2. their last name, Enter first name: Matt Enter last name: Priem Hello Matt Priem! and then outputs a greeting similar to...
-
Given a sorted array 2, 5, 8, 12, 16, 18, 22, 25, 29, 32 and the following interpolation search algorithm. Show the steps of the algorithm when 25 is searched. Also, when 26 is searched. array a,...

Study smarter with the SolutionInn App


