Steady-state evaporation (Fig. 18.2-1), rework the problem solved in S18.2, dealing with the evaporation of liquid A
Question:
Steady-state evaporation (Fig. 18.2-1), rework the problem solved in S18.2, dealing with the evaporation of liquid A into gas B, starting from Eq. 19.1-17.
(a) First obtain an expression for v*, using Eq. (M) of Table 17.8-1, as well as Fick's law in the form of Eq. (D) of Table 17.8-2.
(b) Show that Eq. 19.1-17 then equation: becomes the following nonlinear second-order differentia
(c) Solve this equation to get the mole fraction profile given in Eq. 18.2-11.
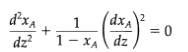
Transcribed Image Text:
(dxA dxA dz? 1- XA ( dz
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
Steadystate evaporation a From Eq M of Table 1781 and the fact that B is stagn...View the full answer

Answered By

Akshay Agarwal
I am a Post-Graduate with a specialization in Finance. I have been working in the Consulting industry for the past 8 years with a focus on the Corporate and Investment Banking domain. Additionally, I have been involved in supporting student across the globe in their academic assignments and always strive to provide high quality support in a timely manner. My notable achievements in the academic field includes serving more than 10,000 clients across geographies on various courses including Accountancy, Finance, Management among other subjects. I always strive to serve my clients in the best possible way ensuring high quality and well explained solutions, which ensures high grades for the students along-with ensuring complete understanding of the subject matter for them. Further, I also believe in making myself available to the students for any follow-ups and ensures complete support and cooperation throughout the project cycle. My passion in the academic field coupled with my educational qualification and industry experience has proved to be instrumental in my success and has helped me stand out of the rest. Looking forward to have a fruitful experience and a cordial working relationship.
5.00+
179+ Reviews
294+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Obtain an expression for the self-inductance per unit length for the parallel wire transmission line of Fig. 5-27(a) in terms of a, d, and , where a is the radius of the wires, d is the axis-to-axis...
-
Obtain an expression for the error term R4 (x) in Problem 57, and find a bound for it if x = 1.2?
-
Obtain an expression for the curve shown in Fig. 7.56 in terms of the LDR and the average normal anisotropy, R.
-
An orange grower in Florida faces a dilemma. The weather forecast is for cold weather, and there is a 50% chance that the temperature tonight will be cold enough to freeze and destroy his entire...
-
A seaplane of total mass m lands on a lake with initial speed vii. The only horizontal force on it is a resistive force on its pontoons from the water. The resistive force is proportional to the...
-
You expect interest rates to rise on five-year bonds by 1 percent per year over the next three years from their artificially low rate of 2 percent. Currently you can buy the following securities at...
-
Derive Eqs. (E.5) and (E.6) of Example 8.10. Data From Example 8.10:- Equation E.5 and E.6:- Determine the effects of rotary inertia and shear deformation on the natural frequencies of a simply...
-
Effects of the level of production on an outsourcing decision Seymour Chemical Company makes a variety of cosmetic products, one of which is a skin cream designed to reduce the signs of aging....
-
Two blocks, joined by a string, have masses of 6.0 and 9.0 kg. They rest on a frictionless horizontal surface. A 2nd string, attached only to the 9-kg block, has horizontal force = 30 N applied to...
-
In the history of the modern Olympics, the United States has won more medals than any other country. But the United States has a large population. Perhaps a better measure of success is the number of...
-
Dehumidification of air (Fig. 19.4-1) for the system of Example 19.4-1 let the vapor is H 2 0 and the stagnant gas is air. Assume the following conditions (which are representative in air...
-
Gas absorption with chemical reaction (Fig. 18.4-1), rework the problem solved in S18.4, by starting with Eq. 19.1-16. What assumptions do you have to make in order to get Eq. 18.4-4?
-
The 2.728-K blackbody radiation has its peak wavelength at 1.062 mm. What was the peak wavelength at t = 700,000 y when the temperature was 3000 K?
-
Given a system consists of eight spin magnetic particles, with applied field B. (a) Show all possible states (i.e. microstates), distinct energy levels of the system, E, and the corresponding...
-
Prove that for fixed t the counting process N(t) has Poisson distribution with parameter at, i.e. N(t) Poisson(at) (Prove that the sum of two exponentially distributed rv-s has gamma distribution,...
-
In this exercise you will see how matrix multiplication can be used to get a nice proof of the major identities that play a role in the extended Euclidean algorithm. We consider two integers a, b e Z...
-
a. Show the income statement (in million $). Expenses without their own specific line listed should go under "All other expenses". Show all your formulas. b. Create a common size income statement (%...
-
The manager of an employee health plan for a firm has studied the balance B, in millions of dollars, in the plan account as a function of t, the number of years since the plan was instituted. He has...
-
If the direction in managerial work today is away from command and control, what is it toward? (a) Coaching and facilitating (b) Telling and selling (c) Pushing and pulling (d) Carrot and stick
-
On January 2, 20X3, Sheldon Bass, a professional engineer, moved from Calgary to Edmonton to commence employment with Acco Ltd., a large public corporation. Because of his new employment contract,...
-
If an auditor becomes aware of a fact that may materially affect the financial report after the date of the auditors report but before the financial report is issued, the auditor should: (a) Discuss...
-
Matters of scale always improve insight and perspective. Reduce the physical size of the gearset in Prob. 1424 by one-half and note the result on the estimates of transmitted load Wt and power.
-
Many years ago a string of Christmas tree lights was manufactured in the form shown in (a). Today the lights are manufactured as shown in (b). Is there a good reason for this change?
-
AGMA procedures with cast-iron gear pairs differ from those with steels because life predictions are difficult; consequently (YN) P, (YN) G, (ZN) P, and (ZN) G are set to unity. The consequence of...
-
Evaluate economic conditions that influence company performance of starbucks Consider political, environmental, currency ( money ) , global economics, and government influences on economic...
-
(Part 1) : The business letter to your employer. The purpose of the letter is to explain why diversity is important in the workplace and to recommend actions the organization can take to become more...
-
Question 4 (20 marks)In the recent annual general meeting of Umi Technology Limited, the board of directors declared an annual dividend of $5.00 per share, and the shareholders agreed to a rights...

Study smarter with the SolutionInn App


