Selected information taken from the financial statements of Ford star Co. for the year ended December 31,
Question:
Selected information taken from the financial statements of Ford star Co. for the year ended December 31, 2022, follows:
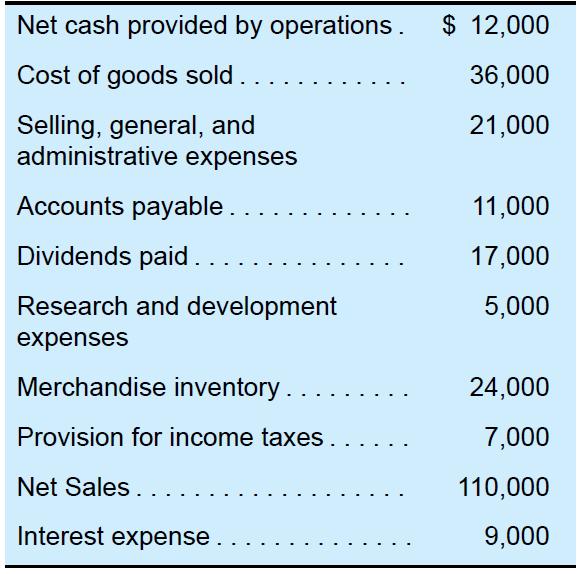
a. Calculate income from operations (operating income) for the year ended December 31, 2022.
b. Calculate net income for the year ended December 31, 2022.
Transcribed Image Text:
Net cash provided by operations. $ 12,000 Cost of goods sold 36,000 21,000 Selling, general, and administrative expenses Accounts payable. Dividends paid.... Research and development expenses Merchandise inventory.. Provision for income taxes.. Net Sales .. Interest expense... 11,000 17,000 5,000 24,000 7,000 110,000 9,000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
a b Net sales Cost of goods sold Gross p...View the full answer

Answered By

Sigei Kipngeno
I am a professional writer with more than 2 years of writing experience. I will always deliver a grade A paper with zero plagiarism . Thanks in advance.
4.70+
175+ Reviews
311+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Business questions
-
Selected information taken from the financial statements of Fordstar Co. for the year ended December 31, 2010, follows: Net cash provided by operations . . . . . . . . . . . . . . . . . . . . . . . ....
-
Selected information taken from the financial statements of Verbeke Co. for the year ended December 31, 2010, follows: Gross profit . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Selected information taken from the financial statements of Maxum Company for two successive years follows. You are to compute the percentage change from 2010 to 2011 whenever possible. Round all...
-
Suppose you have the following training set, and fit a logistic regression classifier : ho(x) = g(00+011+0x2) O O O Which of the following are true? Check all that apply. a) Adding polynomial...
-
Lenny Shafer Bakery and Co. budgeted $ 350,000 to build a factory 10 years from today. Shafer will finance the project by making six equal annual deposits of $ 50,000 at the end of each year...
-
The histograms show triglyceride levels for 87 men and 99 women. Triglycerides are a form of fat found in blood. a. Compare the distributions of triglyceride levels for men and women. (Be sure to...
-
Write a code to test a Gaussian pseudorandom number generator. If you do not have a canned generator available, write a generator based on the Box-Muller algorithm in Appendix I. Apply the following...
-
Inventoriable Costs Presented below is a list of items that may or may not be reported as inventory in a companys December 31 balance sheet. 1. Goods sold on an installment basis (bad debts can be...
-
When a company lends money to employees at a rate of 4%, the company will record ______. Multiple choice question. a liability called Accounts Payable a liability called Notes Payable an asset called...
-
How does gross income differ from income?
-
Selected information taken from the financial statements of Verbeke Co. for the year ended December 31, 2022, follows: a. Calculate income from operations (operating income) for the year ended...
-
A bookkeeper prepared the year-end financial statements of Giftwrap, Inc. The income statement showed net income of $216,000, and the balance sheet showed ending retained earnings of $810,000. The...
-
Use Holt-Winters additive method to create a seasonal model that minimizes the MSE for the data set. Use Solver to determine the optimal values of a, b, and g. a. What are the optimal values of a, b,...
-
1. Discuss new technologies thatshould be implemented today and in the near future to support first responder safety practices. 2.How can the government partner with private industry to improve first...
-
Problem 1: 12 Cabins, 12 Vacancies (4 points) Bates Motel, Inc. is incorporated and headquartered in State Z. It owns and operates several hotels in several states and reports the following...
-
Locate the code of ethics online for Apple. Discuss the impact of the code of ethics on day-to-day operations for Apple? What are the costs of compliance Apple? What are the costs of non-compliance?...
-
Sometimes the governments of free market countries try to control prices to support an industry rather than the functions of supply and demand. For example, the US government supports various...
-
Analyze, and discuss the logistics services listed below and give a practical example that has been practiced in any organization/company/business. You are required to provide as many examples as you...
-
A system is represented by Figure P2.36. (a) Determine the partial fraction expansion and y(t) for a ramp input, r(t) = t, t > 0. (b) Obtain a plot of y(t) for part (a), and find y(t) for t = 1.0 s....
-
A fuel pump sends gasoline from a car's fuel tank to the engine at a rate of 5.88 10-2 kg/s. The density of the gasoline is 735 kg/m3, and the radius of the fuel line is 3.18 10-3 m. What is the...
-
Muscle Beach Inc. makes three models of high-performance weight-training benches. Current operating data are summarized here: Required: a. Calculate the contribution margin ratio of each product. b....
-
Austin Inc. produces small-scale replicas of vintage automobiles for collectors and museums. Finished products are built on a 1/20 scale of originals. The firms income statement showed the following:...
-
HighTech Inc. and OldTime Co. compete within the same industry and had the following operating results in 2019: Required: a. Calculate the breakeven point for each firm in terms of revenue. b. What...
-
Selected accounts of Wildhorse Company are shown as follows. Supplies Expense 7/31 800 Supplies 7/1 Bal. 1,300 7/31 800 7/10 550 Accounts Receivable 7/31 500 Salaries and Wages Expense 7/15 1,500...
-
Propose the structure of the product formed in each of the following reactions. (syntesis) . HCHO Product A HCI H,C 0 NaOH) Product H + H.C PH2 COOH DMF / POCI 3 Product "N" H CH CH HNO AcO Product...
-
d) 3) Use the given information to calculate K for the following reaction and then determine the composition of the reaction mixture: N2(g)+O2(g) 2NO(g) a) 1.6x103; the equilibrium mixture contains...

Study smarter with the SolutionInn App


