A radioactive substance decays at a rate of 18% per year. If the initial amount of the
Question:
A radioactive substance decays at a rate of 18% per year. If the initial amount of the substance is 100 grams, which of the following functions models the remaining amount of the substance, in grams, after t years?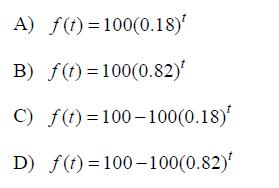
Transcribed Image Text:
A) f(t)=100(0.18)* B) f(t)=100(0.82) C) f(t)=100-100(0.18)* D) f(t)=100-100(0.82)*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
B If the substance decays at a rate of 18 per year the amount of subs...View the full answer

Answered By

Stacy kosgei
I offer quality, original and timely services; Highly credible and void of plagiarism. Your success is my pleasure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Mathematics questions
-
For a simple economy on below table only produce 4 product and services (Book, Burger, Shirts, Cotton). Please note that all cottons will be used in shirts production (cotton isn't final product)....
-
As we saw a radioactive substance decays exponentially: The mass at time t is m(t) = m(0)e kt , where ms0d is the initial mass and k is a negative constant. The mean life M of an atom in the...
-
A sporting event is to be held at the Superdome in New Orleans, Louisiana, which holds about 95,000 people. Suppose 50,000 visitors arrive in New Orleans and spend $500 each. In the month after the...
-
As the company accountant, the management of Nickle Company asks you to explain (a) the concept of reasonable assurance in internal control and (b) the importance of the human factor in internal...
-
The following is Talley Companys income statement for the past year. Sales revenue........ $540,000 Cost of goods sold..... 324,000 Gross margin........ 216,000 Operating expenses..... 126,000...
-
Determine whether the statement is true or false. If it is true, explain why. If it is false, explain why or give an example that disproves the statement. The curve r(t) = (0, t 2 , 4t) is a parabola.
-
Why is it advisable to obtain a written confession from the subject of an investigation?
-
Entries for Asset Acquisition, Including Self-Construction Below are transactions related to Impala Company. (a) The City of Pebble Beach gives the company 5 acres of land as a plant site. The market...
-
You are given the following algorithm written in pseudocode where the entries of the input array A and the input k are positive integer numbers. Also, assume that array A has sufficiently large...
-
The table gives values of f and g at selected values of x. What is the value of f(g(3))? -2 0 3 f(x) -5 6 0 g(x) 0 4 -5
-
If f(x) = 2x and g(x) = 2x 2 , what is the value of f(g(1))-g(f(1))? A) 4 B) 2 C) 2 D) 4
-
For each of the following research projects, list three variables for stratification of the sample. a. A nationwide study of motels and hotels is being conducted. An attempt will be made to determine...
-
Explore the link between construction planning and programming and construction productivity....is their a link? Does good planning and programming improve productivity? Min/Max words 1750 Marking...
-
If you construct a bond portfolio with five 6% coupon (semiannual payment), duration of 2.79 years, 3-year maturity bond sells at par ($1,000) and six 1-year zero coupon bond sells for $950. (a)...
-
1. In the preceding analysis, the estimated safety stock levels are not expressed in integer values. Suppose that inventory levels must be expressed as integers, and re-analyze the case assuming that...
-
Annie purchased a building to use in her trade or business several years ago for $100,000. Annie has previously claimed $25,000 of depreciation on the building. If Annie sells the building for...
-
Morgan Jennings, a geography professor, invests $89,000 in a parcel of land that is expected to increase in value by 13 percent per year for the next eight years. He will take the proceeds and...
-
The following results were obtained at 600 K for the de-composition of ethanol on an alumina (Al2O3) surface C2H5OH(g) C2H4(g) + H2O(g) a. Predict PTotal in torr at t = 80. s. b. What is the value of...
-
Before the 1973 oil embargo and subsequent increases in the price of crude oil, gasoline usage in the United States had grown at a seasonally adjusted rate of 0.57 percent per month, with a standard...
-
Is there any relationship between the type of treatment and the response? What form does the relationship take? Sexually Transmitted Disease Suppose a study examines the relative efficacy of...
-
Suppose either a positive smear or a positive culture is regarded as a positive response and distinguished from the negative smear, negative culture response. Is there an association between the type...
-
What is the appropriate procedure to test whether the rate of DKA is different before and after start of pump therapy? Diabetes Improving control of blood-glucose levels is an important motivation...
-
The IRS suspended the casualty and theft loss deductions for all taxpayers, except those in a federally declared disaster area. What is California's position on this?
-
In 2018, Caterpillar reports cost of goods sold of $36,997 and beginning and ending inventory balances of $10,018 and $11,529. The company's inventory footnote (next slide) provides additional...
-
at its current standard...do you think that the International Property Measurement Standards (IPMS) for all buildings is effective or if it isn't, what does it lack?

Study smarter with the SolutionInn App


