Radium-226 decays with a half-life of 1,620 years to radon-222 plus a helium nucleus ( (alpha) particle)
Question:
Radium-226 decays with a half-life of 1,620 years to radon-222 plus a helium nucleus ( \(\alpha\) particle) according to the nuclear reaction equation
\[{ }_{88}^{226} \mathrm{Ra} ightarrow{ }_{86}^{222} \mathrm{Rn}+{ }_{2}^{4} \mathrm{He} .\]
The atomic masses are

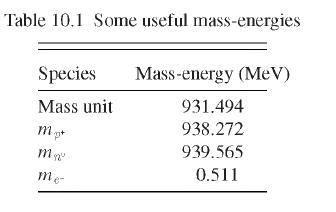
Use the data in Table 10.1 to calculate the energy in \(\mathrm{MeV}\) released in the reaction. How much of this energy appears as kinetic energy of the \(\alpha\) particle?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

An Introduction To Groups And Their Matrices For Science Students
ISBN: 9781108831086
1st Edition
Authors: Robert Kolenkow
Question Posted: