A simple shower for remote locations is designed with a cylindrical tank 500 mm in diameter and
Question:
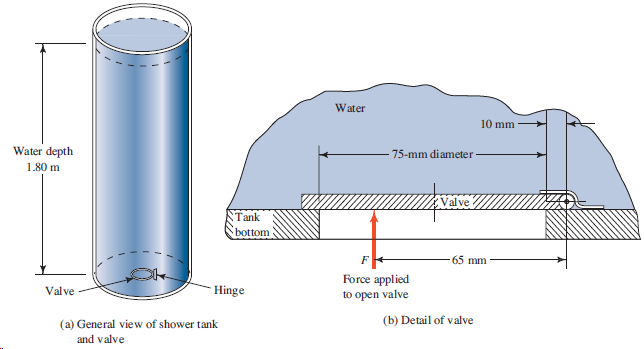
Transcribed Image Text:
Water 10 mm Water depth - 75-mm diameter - 1.80 m Valve Tank bottom 65 mm Force applied to open valve Hinge Valve (b) Detail of valve (a) General view of shower tank and valve
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Force valve pA A 0095 m 2 4 7088 10 3 m 2 ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A cylindrical tank is fully filled with water (Fig. P11??21). In order to increase the flow from the tank, an additional pressure is applied to the water surface by a compressor. For P 0 = 0, P 0 = 3...
-
A cylindrical tank of diameter D contains liquid to an initial height ho. At time t = 0 a small stopper of diameter d is removed from the bottom. Using Bernoullis equation with no losses, derive (a)...
-
A cylindrical tank with hemispherical heads is constructed of steel sections that are welded circumferentially (see figure). The tank diameter is 1.25 m, the wall thickness is 22 mm, and the internal...
-
Nu Company reported the following pretax data for its first year of operations. Net sales 2,930 Cost of goods available for sale 2,420 Operating expenses 750 Effective tax rate 20 % Ending...
-
On December 1, 2017, Papadopoulos SA had the following account balances. During December, the company completed the following transactions. Dec. 7 Received ¬3,600 cash from customers in payment...
-
The file Bulbs contains the life (in hours) of a sample of 40 100-watt light bulbs produced by Manufacturer A and a sample of 40 100-watt light bulbs produced by Manufacturer B. The following table...
-
What are the functions in executing and recording these two classes of transactions and custody of related assets?
-
The management of Style Networks Inc. is considering two TV show projects. The estimated net cash flows from each project are as follows: After Hours requires an investment of $ 913,600, while Sun...
-
1. A long cylindrical rod (Fig. 1) of radius R = 2 cm and length L = 40 cm is made of a material that generates energy from within. The conductivity of the material is k = 15 W/m K. The temperature...
-
A private company has two debt covenants in place: a. Maximum debt- to- equity ratio: Current and long- term liabilities, excluding future income taxes, are divided by total shareholders equity. b....
-
The bottom of a laboratory vat has a hole in it to allow the liquid mercury to pour out. The hole is sealed by a rubber stopper pushed in the hole and held by friction. What force tends to push the...
-
Calculate the total force on the bottom of the closed tank shown in Fig. 4.23 if the air pressure is 52 kPa(gage). Air 0,50 m Oil (sg -0.85) 0.75 m Water 18 m 1.2 m
-
Clark issued $ 50,000 of 10-year, 9% bonds payable on January 1, 2014. Clark pays interest each January 1 and July 1 and amortizes discount or premium by the straight- line amortization method. The...
-
Find a suitable set of state variables, derive the state-variable equations, and form the state equation. \(\frac{1}{3} \dddot{x}+\ddot{x}+\frac{1}{2} \dot{x}+2 x=\cos t\)
-
Find a suitable set of state variables, derive the state-variable equations, and form the state equation. \(m \ddot{x}+b \dot{x}=e^{-2 t / 3}, m, b=\) const \(>0\)
-
Derive the state-variable equations (in vector form) for the given nonlinear system model. \(\left\{\begin{array}{l}\ddot{x}_{1}+\frac{1}{3}\left|\dot{x}_{1} ight| \dot{x}_{1}=x_{2}^{3} \\...
-
Find a suitable set of state variables, derive the state-variable equations, and form the state equation. \(\left\{\begin{array}{l}m L_{1}^{2} \ddot{\theta}_{1}+\left(m g L_{1}+k L_{2}^{2} ight)...
-
Find a suitable set of state variables, derive the state-variable equations, and form the state equation. \(\left\{\begin{array}{l}\ddot{x}_{1}+2 \dot{x}_{1}+2\left(x_{1}-x_{2} ight)=F(t) \\...
-
Do various financial indicators differ significantly according to type of company? Use a one-way ANOVA and the financial database to answer this question. Let Type of Company be the independent...
-
Why is inventory management important for merchandising and manufacturing firms and what are the main tradeoffs for firms in managing their inventory?
-
A system containing argon gas is at pressure P 1 and temperature T 1 . How would you go about estimating the fugacity coefficient of the gas?
-
Is the following statement correct? If not rewrite it so that it is correct. The standard state of water is H 2 O(g).
-
By looking at the a and b values for the van der Waals equation of state, decide whether 1 mole of O 2 or H 2 O has the higher pressure at the same value of T and V.
-
Read this article, then answer the following questions: 1- Description of Instrument: a. Title of instrument, author(s), publication date 2/18/24, 5:43 PM Abuse Risk Inventory for Women: EBSCOhost...
-
According to the article below: 2. What is the hypothesis? TranscribedText: Article Journal of Black Psychology Racial/Ethnic Identity, 2016, Vol. 42(3) 244-258 @ The Author(s) 2015 Religious...
-
Prepare an income statement through gross profit. SPLISH BROTHERS COMPANY Income Statement (Partial) $ $

Study smarter with the SolutionInn App


