Air with a specific weight of 12.5 N/m 3 and a dynamic viscosity of 2.0 Ã 10
Question:
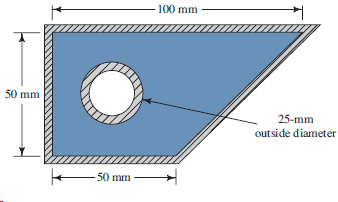
Transcribed Image Text:
100 mm 50 mm 25-mm outside diameter 50 mm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
A 005 m 2 05 005 m 2 0025 m 2 4 3259 10 3 m 2 WP0...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Air with a specific weight of 12.7 N/m 3 and a kinematic viscosity of 1.3 Ã 10 5 m 2 /s is flowing through a flow nozzle similar to that shown in Fig. 15.4. A manometer using water as the gage...
-
A block of wood with a specific weight of 32 lb/ft 3 is 6 by 6 by 12 in. If it is placed in oil (sg = 0.90) with the 6 by 12-in surface parallel to the surface of the oil, would it be stable?
-
(a) If the cone shown in Fig. 5.34 is made of pine wood with a specific weight of 30 lb/ft 3 , will it be stable in the position shown floating in water? (b) Will it be stable if it is made of teak...
-
In Exercises 13 through 24, compute the derivative of the given function and find the equation of the line that is tangent to its graph for the specified value x = c. f(x) = -2 X ; c = - 1
-
What is the difference between an operating line of credit and a bank overdraft?
-
What were the advantages and disadvantages of Roku going public? What were the advantages and disadvantages of going public at the time it chose? Would you recommend that Portland Capital Management...
-
An air standard diesel cycle has a compression ratio of 16. The pressure at the beginning of compression stroke is 1 bar and the temperature is 20C. The maximum temperature is \(1430 C\). Determine...
-
A, B, C, D, and E constituted the board of directors of the X Corporation. While D and E were out of town, A, B, and C held a special meeting of the board. Just as the meeting began, C became ill. He...
-
Which is the most component of the research report?
-
Ann Carter, Chief Financial Officer of Consolidated Electric Company (Con El), must make a recommendation to Con Els board of directors regarding the firms dividend policy. Con El owns two...
-
Carbon dioxide with a specific weight of 0.114 lb/ft 3 and a dynamic viscosity of 3.34 Ã 10 -7 lb-s/ft 2 flows in the shaded portion of the duct shown in Fig. 9.17. If the volume flow rate is...
-
Figure 9.15 shows the cross section of a shell-and-tube heat exchanger. Compute the volume flow rate required in each small pipe and in the shell to obtain an average velocity of flow of 25 ft/s in...
-
Littleton Books has the following transactions during May. May 2 Purchases books on account from Readers Wholesale for $2,300, terms 2/10, n/30. May 3 Pays freight costs of $100 on books purchased...
-
In the dynamic and expanding urban environment of "Slothsberg", a new highway ("Snailpace Highway") is to be constructed over the existing "Dillydally Highway". The following conditions apply to the...
-
This problem explores the relationship between the mass of two blocks and their acceleration. Block A, of mass 2.0 kg, rests on a horizontal tabletop. There is negligible friction between Block A and...
-
Subclass3. Quality if factored into your grades. Students who expend minimal effort on this assignment will not earn full credit. For each class you create, include: At least 3 additional pieces of...
-
A Person Cannot See A Single Cotton Thread 100 Feet Away, But If You Wound Thousands Of Threads Together Into A Rope, It Would Be Visible Much Further Away. Is This Statement Analogous To Our DNA...
-
Given that P(A) = 0.78, P(A|B) = 0.6, and P(A|Bc ) = 0.8. Find P(B) and P(B|A).
-
Figure E19.8 shows ap V-diagram for an ideal gas in which its absolute temperature at bis one-fourth of its absolute temperature at a. (a) What volume does this gas occupy at point b? (b) How many...
-
Arlington Merchants reported the following on its income statement for the fiscal years ending December 31, 2016 and 2015. 2016 2015 Sales $4,857,500 $4,752,900 Cost of goods sold 3,258,950 3,207,000...
-
At 350. K, pure toluene and hexane have vapor pressures of 3.57 10 4 Pa and 1.30 10 5 Pa, respectively. a. Calculate the mole fraction of hexane in the liquid mixture that boils at 350. K at a...
-
The partial molar volumes of water and ethanol in a solution with xH 2 O = 0.45 at 25C are 17.0 and 57.5 cm 3 mol 1 , respectively. Calculate the volume change upon mixing sufficient ethanol with...
-
A solution is made up of 222.9 g of ethanol and 130.8 g of H 2 O. If the volume of the solution is 403.4 cm 3 and the partial molar volume of H 2 O is 17.0 cm 3 mol 1 , what is the partial molar...
-
Diaz Company issued $91,000 face value of bonds on January 1, Year 1. The bonds had a 9 percent stated rate of interest and a ten- year term. Interest is paid in cash annually, beginning December 31,...
-
Manvir had to make payments of $1,125 every 6 months to settle a $22,000 loan that he received at 4.52% compounded semi-annually. a. How long did it take to settle the loan?
-
If I invest a single amount of $14,000 in an account earning 8% p.a. compounding quarterly for 5 years, how much interest will I have earned in those 5 years?

Study smarter with the SolutionInn App


