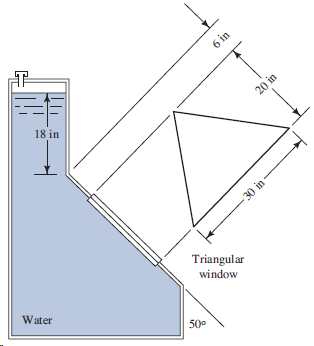
Refer to Fig. 4.41. 6 in 18 in 30 in Triangular window Water 50 20 in
Question:
Transcribed Image Text:
6 in 18 in 30 in Triangular window Water 50° 20 in
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a 18 incos 50 280 in L c a 6 b 280 ...View the full answer

Answered By

Sidharth Jain
My name is Sidharth. I completed engineering from National Institute of Technology Durgapur which is one of the top college in India. I am currently working as an Maths Faculty in one of the biggest IITJEE institute in India. Due to my passion in teaching and Maths, I came to this field. I've been teaching for almost 3 years.
Apart from it I also worked as an Expert Answerer on Chegg.com. I have many clients from USA to whom I teach online and help them in their assignments. I worked on many online classes on mymathlab and webassign. I guarantee for grade 'A'.
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Refer to Fig. 4.33. Water 8 ft Access hatch for cleaning 30 in 3 ft 45 18 in 18 in 18 in
-
Refer to Fig. 4.34. 6-in diameter 3 ft 2 ft 1 ft View port Oil (sg -0.90) 30
-
Refer to Fig. 4.35. 0.6 m Oil (sg - 0.90) +300 Window 0,6 m dimen sions in mm 0.3 m 40 -1.2 m + 300+300
-
1. You are the audit partner in charge of five audits which have recently been completed. You are reviewing the files and you now only have to decide which form of audit report you will sign off....
-
Nestlé's financial statements are presented in Appendix B. Financial statements of Petra Foods are presented in Appendix C. Instructions (a) Based on the information contained in these...
-
In Problem 9.52, if the null hypothesis is that 20% of the items in the population are defective, what is the value of ZSTAT?
-
Identify the departments that report to the (a) vice-president of sales, (b) vice-president of finance, and (c) controller.
-
Teresa Ramirez and Lenny Traylor are examining the following statement of cash flows for Pacific Clothing Stores first year of operations. Teresa claims that Pacifics statement of cash flows is an...
-
2. [50 pts] Using microarray technology, an estimate of a gene's expression in a cohort of subjects can often be approximated as normally distributed because the gene's expression is often averaged...
-
Ezzell Enterprises has the following capital structure, which it considers to be optimal under present and forecasted conditions: Debt (long-term only) ........ 45% Common equity .......... 55 Total...
-
What is an SQL injection attack and how can it be prevented?
-
Figure 4.42 shows a gasoline tank filled into the filler pipe. The gasoline has a specific gravity of 0.67. Calculate the total force on each flat end of the tank and determine the location of the...
-
Suppose you discovered that on your college campus, 6% of the female students were married and 4% of the female students had at least one child. a. From this information, is it possible to determine...
-
Define virtual visitation. Give an example.
-
Describe how the majority of courts treat the existence of multiple families when making decisions about child support.
-
Identify some of the ways in which courts address parental responsibility for the cost of their childrens higher education.
-
Identify the kinds of objections to discovery requests that can be raised.
-
Describe the nature and purpose of financial statements/affidavits.
-
Compute (a) The characteristic polynomial of A, (b) The eigenvalues of A, (c) A basis for each eigenspace of A, (d) The algebraic and geometric multiplicity of each eigenvalue. 1 0 0 A = 1 1 3 -2 1 -1
-
9.Consider the reaction 3NO2(g)+H2O=2HNO3(aq)+NO(g) where Delta H=-137 kJ.How many kilojoules are released when 92.3g of NO2 reacts?
-
Benzoic acid, 1.35 g, is reacted with oxygen in a constant volume calorimeter to form H 2 O(l) and CO 2 (g) at 298 K. The mass of the water in the inner bath is 1.55 10 3 g. The temperature of the...
-
Calculate the P and T values for which Br2(g) is in a corresponding state to Xe(g) at 330. K and 72.0 bar.
-
For each of the following reactions, predict the product and draw the mechanism of its formation. a. b. c. d. e. f. 1) PhMgBr 2) H20 Me 1) NaCN 2) H20 *Me
-
2. After scouting a number of locations, your financial analysts have settled on a desirable location for one of the firm's new manufacturing plants. However, the state income and local property...
-
The "one-time good deal" Cash-For-Clunkers program offered by the federal government proved a temporary boon for car dealers. In addition to this program, dealers were eager to add their own...
-
1. You are Chief Financial Officer for a company that sells contact lenses by mail order to a notional market. Despite the national presence, the firm's facilities are all located in one state. Your...

Study smarter with the SolutionInn App


