To what height will the jet of water rise for the conditions shown in Fig. 6.40? p=
Question:
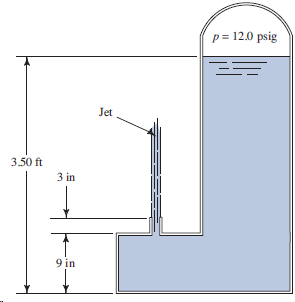
Transcribed Image Text:
p= 12.0 psig Jet 3.50 ft 3 in 9 in
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
h 350 ft 10 ft 250 ft D...View the full answer

Answered By

Muqadas Javed
I am a mentor by profession since seven years. I have been teaching on online forums and in universities. Teaching is my passion therefore i always try to find simple solution for complicated problems or task grasp them so that students can easily grasp them.I will provide you very detailed and self explanatory answers and that will help you to get good grade. I have two slogans: quality solution and on time delivery.
4.60+
24+ Reviews
144+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
To what height will the jet of fluid rise for the conditions shown in Fig. 6.39? Jet 2.60 m 75 mm 0.85 m
-
A glass rod of diameter d1 = 1.5 mm is inserted sym- metrically into a glass capillary with inside diameter d2 = 2.0 mm. Then the whole arrangement is vertically oriented and brought in contact with...
-
Water of mass m = 20 g is enclosed in a thermally insulated cylinder at the temperature of 0 C under a weightless piston whose area is S = 410 cm2 . The outside pressure is equal to standard...
-
Select only 2. Two questions which can be used to address ethical issues impacting organizations and Human Resource Management include: What would my Supervisor say? Does this behavior or result meet...
-
Journalize, without explanations, the following transactions of Digbey Auto Parts, a distributor that uses the periodic inventory system, during the month of June 2017: Jun. 3 Purchased $16,800 of...
-
Thomas Industries has the following patents on its December 31, 2014, balance sheet. The following events occurred during the year ended December 31, 2015. 1. Research and development costs of...
-
Motion Auto has the following information for the years ending December 31,2010 and 2009: Requirements 1. Compute the rate of inventory turnover for Motion Auto for the years ended December 31, 2010...
-
Pool Corporation, Inc., reported in its recent annual report that In 2010, our industry experienced some price deflation. . . . In 2011, our industry experienced more normalized price inflation of...
-
Good day dear tutor, please answer all the requirements , I deeply appreciat it thank you so much! Financial mix ratios. The data given below were obtained from the financial records of Menace V....
-
Lina purchased a new car for use in her business during 2022. The auto was the only business asset she purchased during the year, and her business was extremely profitable. Calculate her maximum...
-
What pressure is required above the water in Fig. 6.12 to cause the jet to rise to 28.0 ft? The water depth is 4.50 ft. h
-
Solve Problem 6.88 using the direct application of torricelli's theorem. What depth of fluid above the outlet nozzle is required to deliver 200 gal/min of water from the tank shown in Fig. 6.37? The...
-
On October 1, 2023, Ross Wind Energy Inc. issued a $1,500,000, 7%, seven-year bond. Interest is to be paid annually each October 1. Required a. Calculate the issue price of the bond assuming a market...
-
Identify the different ways the polluter-pays principle is given effect in international law.
-
Discuss the relationship between the polluter-pays principle and the principles of prevention and precaution.
-
Critically assess the roles of the International Court of Justice and the International Tribunal for the Law of the Sea in being a competent forum for settling environmental disputes.
-
What are the differences between the regimes for state responsibility and civil liability for environmental harm?
-
Discuss the problems of high seas fishing.
-
Which of the following is a valid SQL command? a. ALTER TABLE books ADD CONSTRAINT UNIQUE (pubid); b. ALTER TABLE books ADD CONSTRAINT PRIMARY KEY (pubid); c. ALTER TABLE books MODIFY (pubid...
-
Velshi Printers has contracts to complete weekly supplements required by fortysix customers. For the year 2018, manufacturing overhead cost estimates total $600,000 for an annual production capacity...
-
When used as a solvent, will acetone favor an S N 2 or an S N 1 mechanism? Explain. Acetone
-
Determine whether each of the following reactions proceeds via an S N 1 or S N 2 mechanism and then draw the product(s) of the reaction: (a) (b) (c) (d) (e) (f) .? , Br cP HMPA
-
In Chapter 23, we will learn several methods for making primary amines (RNH 2 ). Each of these methods utilizes a different approach for forming the CN bond. One of these methods, called the Gabriel...
-
Lightfoot Inc., a software development firm, has stock outstanding as follows: 32,000 shares of cumulative preferred 1% stock, $145 par and 107,000 shares of $160 par common. During its first four...
-
A student is working with a 3.00-F capacitor, a 5.75-uF capacitor, and a 6.40-V battery. What is the the charge (in C) on each capacitor if the student connects the capacitors in the following ways....
-
1. Here is a graph of the distance for another airplane trip. Mark where the velocity is fast and where it is slow. (You can think of the "fast" periods of time as times when the plane enjoys a...

Study smarter with the SolutionInn App


