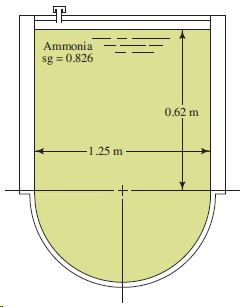
Use Fig. 4.48. The surface is 2.50 m long. Ammonia sg = 0.826 0,62 m 1.25 m
Question:
Transcribed Image Text:
Ammonia sg = 0.826 0,62 m 1.25 m
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
F v V Aw F v 0826 981 kNm 3 1389 ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use Fig. 4.47. The surface is 2.00 m long. Water 1.85 m 0.75-m radius
-
Use Fig. 4.53. The surface is 1.50 m long. 2.80 m 1.20 m +: Water
-
Use Fig. 4.54. The surface is 60 in long. 48 in 36 in Alcohol sg = 0.79
-
Solve the problem. Jake Dowell has total fixed monthly expenses of $1367 and his gross monthly income is $3861. What is his debt-to-income ratio? Round to the nearest percent. ? 3% o 4% o 28% o 35%
-
Zhu Boards sells a snowboard, Xpert that is popular with snowboard enthusiasts. Information relating to Zhu's purchases of Xpert snowboards during September is shown on the next page. During the same...
-
Zagat's publishes restaurant ratings for various locations in the United States. The file Restaurants contains the Zagat rating for food, decor, service, and the cost per person for a sample of 100...
-
What methods are used in (a) the preliminary review and (b) the completion of the review.
-
Manning Corporation is considering a new project requiring a $ 90,000 investment in test equipment with no salvage value . The project would produce $ 66,000 of pretax income before depreciation at...
-
1 A B D E F G H Part One 2 Prepare a perpetual inventory record using the UFO method for each of Sterling Cutlery's three different products K L M N P a R LIFO Method 3 4 Item # 1 5 Date 6 Quantity...
-
Design the psuedocode logic for a program that reads sales data from sales.dat file and calculate the total sales and the average of sales, and then display the results. Assume that the sales.dat...
-
Repeat Problem 4.28 (Fig. 4.40), except that the tank is now sealed at the top with a pressure of 4.0 psig above the fluid. 8.0 in Semicircular hatch 10 in 26 in 20-in radius Ethylene glycol sg =...
-
Use Fig. 4.49. The surface is 5.00 ft long. Water 10.00 ft 75 15.00 ft
-
What is the value of the equilibrium constant for the dissociation of acetic acids proton? What does this value tell you?
-
If the Australian Mining Corporation earned A\($550,000\) (Australian dollar) in net income, spent A\($110,000\) in cost of goods sold, had A\($45,000\) of general and administrative expenses,...
-
Define and give two examples of extraordinary expenses. How do the courts generally deal with such expenses?
-
What would you expect the nominal rate of interest of German government bonds to be if the real rate is 6 percent and the expected inflation rate is 9 percent?
-
Go to the Wall Street Journal home page at www.wsj.com. Search for any firm of your choice. Find its earnings announcements. What did you learn about the company from the announcements? Also, search...
-
Describe the paralegals role in a property division case.
-
Compute the determinants using cofactor expansion along any row or column that seems convenient. al |0 d e h i j
-
Stephen Schor, an accountant in New York City, advised his client, Andre Romanelli, Inc., to open an account at J. P. Morgan Chase Bank, N.A., to obtain a favorable interest rate on a line of credit....
-
Predict the product of each of the following reactions: (a) (b) [H2SO4] excess MeOH -? -H20 [H,SO4] -H20
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OH
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) OH H.
-
2. You are tasked with identifying a piece of metal that has either a BCC or FCC structure. The following x-ray diffraction spectra using Cu-Ka radiation (0.1542 nm) corresponds to the metal. Use the...
-
In March, Roland had revenues of $612,000 fixed costs of $199,260, and a profit of $51,660. Answer the following questions: Required: a. What was the contribution margin ratio? b. What monthly sales...
-
Tinder Confections produces fudge truffles in 10-pound batches. Each batch takes 0.15 hours of direct labor, which includes allowances for breaks, cleanup, and other downtime. Tinder pays its direct...

Study smarter with the SolutionInn App


