The table measures the altitude of a falling parachutist before her chute has opened. Predict how many
Question:
The table 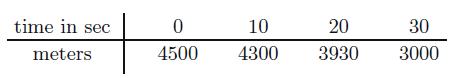 measures the altitude of a falling parachutist before her chute has opened. Predict how many seconds she can wait before reaching the minimum altitude of 1500 meters.
measures the altitude of a falling parachutist before her chute has opened. Predict how many seconds she can wait before reaching the minimum altitude of 1500 meters.
Transcribed Image Text:
time in sec meters 0 4500 10 4300 20 3930 30 3000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
ANSWER To predict how many seconds the parachutist can wait before reaching the minimum altitude of ...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Mathematics questions
-
A psychologist interested in nonverbal communication decided to do an informal study on eye contact. Knowing that eye contact tends to be associated with an emotional connection, she believed that...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Diagonalize the matrices, if possible. The eigenvalues are as follows: (11) = 1, 2, 3; (12) = 1, 4; (13) = 5, 1; (14) = 3, 4; (15) = 3, 1; (16) = 2, 1. 3 4 6 1
-
What was the basis for inflation during World Wars I and II?
-
The theoretical cycle time for a product is 30 minutes per unit. The budgeted conversion costs for the manufacturing cell are $2,700,000 per year. The total labor minutes available are 600,000....
-
What is a System Maintenance Concept? Identify examples of its UCs.
-
Sterling Steel Inc. purchased a new stamping machine at the beginning of the year at a cost of $580,000. The estimated residual value was $60,000. Assume that the estimated useful life was five...
-
In January 2016, Starbucks filed a lawsuit against the parent company of New York's Coffee Culture Cafe for launching a drink called the "Freddocino" The lawsuit's documents allege that not only does...
-
The management of a certain chain of supermarkets would like to know on a comparative basis the performance of its supermarkets in each city. The data in the following table shows the amount of...
-
A missile is launched in your direction. Using a range finder, you measure its altitude at the times: How long until you have to run? time in sec altitude in meters 0 200 10 650 20 30 40 970 1200...
-
Let b = (0, 3, 1, 2) T . Find the vector w span {(0, 0, 1, 1) T , (2, 1, 1,1 ) T } such that w * b is minimized.
-
In Problem use the table below. Events A, B, and C are mutually exclusive; so are D, E, and F. In Problem test each pair of events for independence. D and F
-
The virial equation of state is \[p v=\Re T\left(\mathrm{~b}_{1}+\frac{\mathrm{b}_{2}}{v}+\frac{\mathrm{b}_{3}}{v^{2}}+\ldots . . ight)\] Compare this equation with van der Waals equation of state...
-
A vessel is filled with hydrogen and carbon dioxide in equal parts by volume and the mixture is ignited. If the initial pressure and temperature are \(2 \mathrm{bar}\) and \(60^{\circ} \mathrm{C}\)...
-
A mixture containing hydrogen and oxygen in the ratio of \(2: 1\) by volume is contained in a rigid vessel. This is ignited at \(60{ }^{\circ} \mathrm{C}\) and a pressure of \(1 \mathrm{~atm}(1.013...
-
The exhaust gas from a two-stroke cycle compression-ignition engine is exhausted at an elevated pressure into a large chamber. The gas from the chamber is subsequently expanded in a turbine. If the...
-
The following data refer to an analysis of a dual combustion cycle with a gas having specific heats varying linearly with temperature: The pressure and temperature of the gas at the end of...
-
Does the statement of Clausius require a constant T for the heat transfer as in a Carnot cycle?
-
Identify the Critical Infrastructure Physical Protection System Plan.
-
(a) Determine whether is in the span of (b) Is In the span of and (c) Is in the span of and 123 011 110 121 120 3 3012 0130 1201 2011
-
Prove parts (b) and (c) of Theorem 2.21. Theorem 2.21 (b) The vectors are linearly independent if and only if the only solution to the homogeneous system Ac = 0 is the trivial one, c = 0. (c) A...
-
(a) Prove that if v1,.......vn are linearly independent, then any subset, e.g., v1.......vk with k < m, is also linearly independent. (b) Does the same hold true for linearly dependent vectors? Prove...
-
For the first seven questions, consider the economy of Columbia. In this economy, there are three firms potentially interested in issuing a $100,000 bond to do a project each hopes will be...
-
Find the average percentage (%) earned on retail loans: Net loans are $731,266 at the end of 2012, interest income earned on net loans is $30,522. 4.4% of net loans are retail loans. What is the...
-
Compute cost of goods sold using the following information. Finished goods inventory, beginning Work in process inventory, beginning Work in process inventory, ending Cost of goods manufactured...

Study smarter with the SolutionInn App


