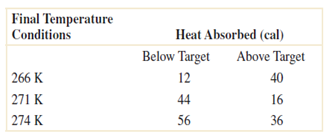
Consider the endothermic reactions given below. Let A denote the event that a reaction's final temperature is
Question:
Transcribed Image Text:
Final Temperature Conditions Heat Absorbed (cal) Below Target Above Target 266 K 12 40 271 K 44 16 274 K 56 36
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
PA 112204 05490 PB 92204 ...View the full answer

Answered By

Cristine kanyaa
I possess exceptional research and essay writing skills. I have successfully completed over 5000 projects and the responses are positively overwhelming . I have experience in handling Coursework, Session Long Papers, Manuscripts, Term papers, & Presentations among others. I have access to both physical and online library. this makes me a suitable candidate to tutor clients as I have adequate materials to carry out intensive research.
4.90+
1538+ Reviews
3254+ Question Solved
Related Book For 

Applied Statistics And Probability For Engineers
ISBN: 9781118539712
6th Edition
Authors: Douglas C. Montgomery, George C. Runger
Question Posted:
Students also viewed these Mathematics questions
-
Consider the endothermic reactions given below. Let A denote the event that a reaction's final temperature is 271 K or less. Let B denote the event that the heat absorbed is above target. Determine...
-
Consider the endothermic reactions given below. Let A denote the event that a reaction's final temperature is 271 K or less. Let B denote the event that the heat absorbed is above target. Use the...
-
Consider the endothermic reactions given below. Use Bayes theorem to calculate the probability that a reaction's final temperature is 271 K or less given that the heat absorbed is above target. Final...
-
In Exercises use the Trapezoidal Rule and Simpson's Rule to approximate the value of the definite integral for the given value of n. Round your answer to four decimal places and compare the results...
-
Consider the homeowners association presented at the beginning of this section. A random sample of 20 households resulted in 15 indicating that they would favor an increase in assessments. Explain...
-
Two products are obtained from the reaction of (Z)-2-pentene with water and a trace of H2SO4. The mass spectra of these products are shown in Figure 13.10. Identify the compounds responsible for the...
-
With the rider in the racing position, how much more power is required to pedal a bicycle at \(15 \mathrm{mph}\) into a \(20-\mathrm{mph}\) head-wind than at \(15 \mathrm{mph}\) through still air?...
-
(a) Generate the material requirements. You need a BOM for each of the three products (A, B, and C), beginning inventory levels, and scheduled receipts. The BOMs are shown in Figure. All items use...
-
StorSmart Company makes plastic organizing bins. The company has the following inventory balances at the beginning and end of March: S Raw materials Work in process Finished goods Beginning Inventory...
-
Given a string, reduce it in such a way that all of its substrings are distinct. To do so, you may delete any characters at any index. What is the minimum number of deletions needed? Note: A...
-
Consider the well failure data given below. Let A denote the event that the geological formation has more than 1000 wells, and let B denote the event that a well failed. Are these events independent?...
-
In an acid-base titration, a base or acid is gradually added to the other until they have completely neutralized each other. Because acids and bases are usually colorless (as are the water and salt...
-
Synthesize each of the following compounds by routes that involve both allylic and benzylic bromination by NBS. (a) (b)
-
What is the coupon rate of a 9-year,$1,000bond with coupons paid annually and a price of $900,if it has a yield to maturity of 5.00%? NOTE:Answer with two decimals after the dot.DO NOT USE the...
-
For each transaction, indicate in which journal it should be recorded. Sales Journal Cash Receipts Journal Purchases Journal Cash Payments Journal General Journal Returned products to a supplier....
-
For each charge distribution: -6.0 x 105 C Z 9 E 2.0 m - 6.0 x 10-5 C 3.0 m 4.0 x 105 C A 92 3.0 m q2 2.0 m 2 -3.0 x 10-5 C A a. Determine the net electric field at point A. b. The magnitude of the...
-
An institution has just sold a European put option contract on 600,000 British pounds. The strike price of the option is 1.26 and the time to maturity is 26 weeks. The current exchange rate for...
-
Re-write the following, eliminating "legalese, No later than 90 days after the end of each fiscal year, the Borrower shall provide the Bank a copy of the Borrower's year-end financial statements....
-
In problem, use the given function f to: (a) Find the domain of f. (b) Graph f. (c) From the graph, determine the range and any asymptotes of f. (d) Find f -1 , the inverse of f. (e) Find the domain...
-
Frontland Advertising creates, plans, and handles advertising campaigns in a three-state area. Recently, Frontland had to replace an inexperienced office worker in charge of bookkeeping because of...
-
A farmer interested in the weight of his soybean crop randomly samples 100 plants and weighs the soybeans on each plant? A research objective is presented. For each, identify the population and...
-
Every year the U.S. Census Bureau releases the Current Population Report based on a survey of 50,000 households. The goal of this report is to learn the demographic characteristics, such as income,...
-
Contrast the differences between qualitative and quantitative variables?
-
What innovative approaches can organizations employ to reconcile competing demands for capital investment and operational expenditure within the constraints of finite budgetary resources, while still...
-
What measures can organizations implement to promote transparency and accountability in the budgeting process, thereby fostering trust and buy-in among stakeholders and facilitating...
-
Based on the following cost expenditures for the construction of an office / warehouse development ( to include direct and indirect charges ) , calculate the peak financial requirement: Month Monthly...

Study smarter with the SolutionInn App


