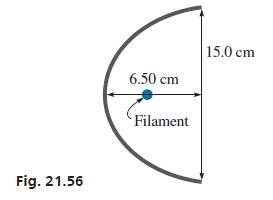
A spotlight with a parabolic reflector is 15.0 cm wide and is 6.50 cm deep. See Fig.
Question:
A spotlight with a parabolic reflector is 15.0 cm wide and is 6.50 cm deep. See Fig. 21.56 and Example 4. Where should the filament of the bulb be located so as to produce a beam of light?
Transcribed Image Text:
Fig. 21.56 6.50 cm Filament 15.0 cm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
To determine the location of the filament of the bulb we need to find the focal point ...View the full answer

Answered By

FELIX NYAMBWOGI
I have been tutoring for over 5 years, both in person and online. I have experience tutoring a wide range of subjects, including math, science, English, and history. I have also worked with students of all ages, from elementary school to high school.
In addition, I have received training in effective tutoring strategies and techniques, such as active listening, questioning, and feedback. I am also proficient in using online tutoring platforms, such as Zoom and Google Classroom, to effectively deliver virtual lessons.
Overall, my hands-on experience and proficiency as a tutor has allowed me to effectively support and guide students in achieving their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Basic Technical Mathematics
ISBN: 9780137529896
12th Edition
Authors: Allyn J. Washington, Richard Evans
Question Posted:
Students also viewed these Mathematics questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The filament of a light bulb is a thin wire that glows when electricity passes through it. The filament of a car headlight is at the focus of a parabolic reflector, which sends light out in a...
-
A researcher wanted to find out if there was difference between older movie goers and younger movie goers with respect to their estimates of a successful actors income. The researcher first...
-
Refer to the data for Alou Equipment Repair Corp. in P4.9A. Assume that Alou closes its books monthly. In P4.9A On October 31, 2018, Alou Equipment Repair Corp.'s opening trial balance was as...
-
A small company has the technology to develop a new personal data assistant (PDA), but it worries about sales in the crowded market. They estimate that it will cost $600,000 to develop, launch, and...
-
Nozzles and diffusers are widely used in (a) Heat exchangers (b) Refrigeration systems (c) Rockets and other space vehicles (d) None of these.
-
Using one beam element and one spring element, find the natural frequencies of the uniform, spring-supported cantilever beam shown in Fig. 12.22. Figure 12.22:- p. A,I,E X X+ 7777 b Section X-X h...
-
The accounting department of a large limousine company is analyzing the costs of its services. The cost data and level of activity for the past 16 months follow: In addition to the above information,...
-
calculate sale proceeds use those information below oard Name of Investment Purchase Price Indie Ruth Asset $50,000,000 Date Acquired 1/3/2021 Sources of Capital $ Amount Ratio % Debt Proceeds $0 0%...
-
A radio signal is sent simultaneously from stations A and B 600 km apart on the Carolina coast. A ship receives the signal from A 1.20 ms before it receives the signal from B. Given that radio...
-
An airplane wing is designed such that a certain cross section is an ellipse 8.40 ft wide (horizontally) and 1.20 ft thick (vertically). Find the equation of this ellipse if the center is at the...
-
Firms can control their accruals within fairly wide limits. Discuss.
-
Determine the income tax consequences of the share redemption to the major shareholder. Start by calculating the taxable non-eligible dividend. Proceeds of Redemption Less: Paid-up Capital ITA 84(3)...
-
The equilibrium constant, Kc, for the following reaction is 1.2910-2 at 600 K. CoCl2(g) CO(g) + Cl2(g) Calculate the equilibrium concentrations of reactant and products when 0.323 moles of COCl2(g)...
-
For each of the unrelated transactions described below, present the entries required to record each transaction. 1. 2. 3. Oriole Corp. issued $20,300,000 par value 10% convertible bonds at 98. If the...
-
What is the relationship between traditional security issues and human security issues? Can you think of an example of a human security issue that has occurred in your city?
-
1. describe the scope of Public Sector Accounting? Review the topics in the public sector course and what about the government in Indonesia??? 2. Explain the role of accounting in realizing good...
-
Chandler Burford owns a masonry home located in zone 2. What is the annual homeowners insurance premium if the home is insured for $350,000, the owner has an OCC credit rating and chooses a...
-
Wilsons Auto Repair ended 2011 with Accounts Receivable of $85,000 and a credit balance in Allowance for Uncollectible Accounts balance of $11,000. During 2012, Wilsons Auto Repair had the following...
-
Gatorade Use the following table, which shows the amount of sugar, in grams (g), and caffeine, in milligrams (mg), in an 8-oz serving of selected beverages. Let the beverages listed represent the...
-
Use the Venn diagram in Fig. 2.24 to list the sets in roster form. A' - B 12 10 4 6 \14 6. 11 2 13
-
Use the Venn diagram in Fig. to list the set of elements in roster form. A ª B' (2 4 10 11
-
What societal critiques does Virginia Woolf present through her experimental narrative structure and exploration of gender roles in "Mrs. Dalloway"?
-
You are a summer clerk at a law firm in Tucson. The senior partner at the firm calls you into her office to discuss a new case that she would like you to work on. The firm is taking the case pro bono...
-
What is the most important or interesting fact. Write a focus sentence identifying that newsworthy angle: 1. The New England Journal of Medicine released a study today. The study say people who...

Study smarter with the SolutionInn App


