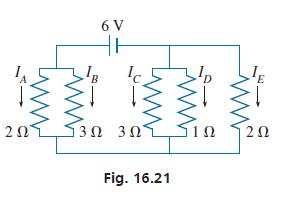
In applying Kirchhoffs laws to the circuit shown in Fig. 16.21, the following equations are found. Determine
Question:
In applying Kirchhoff’s laws to the circuit shown in Fig. 16.21, the following equations are found. Determine the indicated currents (in A).
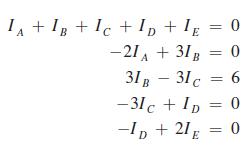
Transcribed Image Text:
IA +IB +IC+ID+1E = 0 -21A +31B = 0 31B - 31 c = 6 -31c + ID 0 C -ID + 21E = 0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
The circuit shown in Fig 1621 can be analyzed using Kirchhoffs laws Applying Kirchhoffs first law at ...View the full answer

Answered By

Mary Boke
I have teached the student upto class 12th as well as my fellow mates.I have a good command in engineering,maths and science.I scored 90+ marks in 10th and 12th in maths.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Basic Technical Mathematics
ISBN: 9780137529896
12th Edition
Authors: Allyn J. Washington, Richard Evans
Question Posted:
Students also viewed these Mathematics questions
-
Evaluate the line intigral I=[(x-8) ds aheve the semicircle of Tadies from A= (a, 0) to B=(-a,0) and for ahieh y20 is numing a
-
In applying Kirchhoffs laws to the circuit shown in Fig. 16.15, the following equations are found. Determine the indicated currents (in A). Fig. 16.15. Ic IA + B + IC = 0 = 6 21 A 51 B 51B - Ic = -3
-
Apply Kirchhoffs laws to the circuit shown in Figure 9.15 to determine the current that will be shown by the ammeters A 1 , A 2 and A 3 . 20 (A1 10 V (A2 202 5.0 V (A3) Figure 9.15: Kirchhoff's laws...
-
How to get a good answer using data and analysis tools in jmp 1 uses the other variables to predict "passion." passion would need to be aggregated into high/low first, and then you can use logistic...
-
Using the molecular orbital model to describe the bonding in CO, CO+, and CO2+, predict the bond orders and the relative bond lengths for these three species. How many unpaired electrons are present...
-
In several cases, a nearby star has been found to have a large planet orbiting about it, although the planet could not be seen. Using the ideas of a system rotating about its center of mass and of...
-
When auditing within the expenditure/disbursement cycle, auditors often rely predominantly on interim tests of controls rather than year end substantive tests of details. In fact, in many cases, year...
-
Rhince and Rynelf decide to merge their proprietorships into a partnership calledDawn Treader Company. The balance sheet of Rynelf Co. shows: The partners agree that the net realizable value of the...
-
When you can draw from your own experiences, including past conversations, you will be less inclined to "overthink" it, as a multimodal learner might be inclined to do. So it's important, as you...
-
Solve the given systems of equations using the inverse of the coefficient matrix. 5A 7B = 62 6A + 5B = 6
-
Find the indicated matrices using a calculator. 6B 4A A = c = C 6-3 4 -5 B = -1 4 -7 2 -6 11 3 -9 D = 12 -6 79-6 -4 0 8
-
1. An IT auditor is evaluating their company's system development and change management process. Which of the following would provide the GREATEST concern to the IT auditor? i. The project was over...
-
Discuss the best promotional effort y have ever experienced. It can be an example from any aspect of the promotional mix. Describe what it is and why you found it to be so effective. If possible,...
-
Beginning inventory Merchandise Finished goods Cost of merchandise purchased Cost of goods manufactured Ending inventory Merchandise Finished goods Unimart Bare Manufacturing $ 316,000 $ 632,000...
-
Castile Incorporated had a beginning balance of $2,100 in its Accounts Receivable account. The ending balance of Accounts Receivable was $2,800. During the period, Castile recognized $55,000 of...
-
Novak Inc. issued $3,840,000 of 11%, 10-year convertible bonds on June 1, 2025, at 99 plus accrued interest. The bonds were dated April 1, 2025, with interest payable April 1 and October 1. Bond...
-
Steph and Maya are standing atop a building looking down at a swimming pool. The pool is 10 m away from the building, and the building is 25 m tall. To jump off the pool, Maya tells Steph to run at 5...
-
What are the different components of foreign investment? Why has the distinction between them begun to blur in recent years?
-
Jax Incorporated reports the following data for its only product. The company had no beginning finished goods inventory and it uses absorption costing. $ 57.30 per unit $ 10.30 per unit $ 7.80 per...
-
The regular price of a Phillips color TV is $539.62. During a sale, Hill TV is selling the TV for $439. Determine the percent decrease in the price of this TV. Where appropriate, round your answer to...
-
On January 1, Juan weighed 235 pounds and decided to diet and exercise. On June 30, Juan weighed 210 pounds. Determine the percent decrease in Juans weight from January 1 to June 30. Where...
-
The cost of a soccer ball to Soccer Line Sports Goods is $35. They sell the same soccer ball for $49. Determine the percent markup in the price of the soccer ball. Where appropriate, round your...
-
I like to think that I have been quite lucky when it comes to working under great leadership. The one that sticks out the most is the police lieutenant I worked under at a community college campus...
-
Given $$ z(x, y)-\sin\left(x^{4} y^{2} ight) $$ find $\frac{\partial z}{\partial y}$. Enter your answer below as a function of $x$ and $y$ : ?
-
Regarding the diagnostic/ analytic models appropriate for analyzing the data, either an individual-level diagnostic model or an organization-level diagnostic model would work. Briefly explain how...

Study smarter with the SolutionInn App


