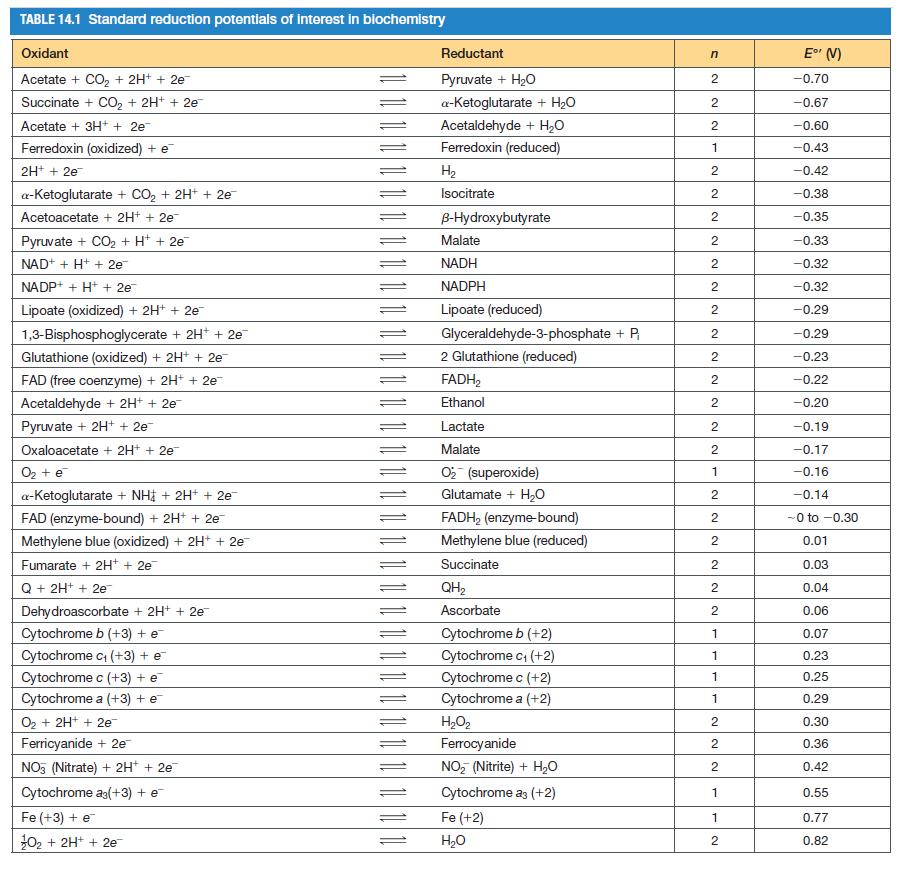
From E values in Table 14.1, calculate the equilibrium constant for the glutathione peroxidase reaction at 37
Question:
From E°′ values in Table 14.1, calculate the equilibrium constant for the glutathione peroxidase reaction at 37 °C.
Table 14.1
Transcribed Image Text:
TABLE 14.1 Standard reduction potentials of interest in biochemistry Oxidant Acetate + CO₂ + 2H+ + 2e¯ Succinate + CO₂ + 2H+ + 2e- Acetate + 3H+ + 2e¯ Ferredoxin (oxidized) + e 2H+ + 2e a-Ketoglutarate + CO₂ + 2H+ + 2e¯ Acetoacetate + 2H+ + 2e™ Pyruvate + CO₂ + H+ + 2e™ NAD+ + H+ + 2e™ NADP+ + H+ + 2e Lipoate (oxidized) + 2H+ + 2e- 1,3-Bisphosphoglycerate + 2H+ + 2e™ Glutathione (oxidized) + 2H+ + 2e FAD (free coenzyme) +2H+ + 2e™ Acetaldehyde + 2H+ + 2e Pyruvate + 2H+ + 2e Oxaloacetate + 2H+ + 2e O₂ + e a-Ketoglutarate + NH + 2H+ + 2e FAD (enzyme-bound) + 2H+ + 2e™ Methylene blue (oxidized) + 2H+ + 2e Fumarate + 2H+ + 2e¯ Q + 2H+ + 2e™ Dehydroascorbate + 2H+ + 2e Cytochrome b (+3) + e Cytochrome c₁ (+3) + e Cytochrome c (+3) + e Cytochrome a (+3) + e 0₂ + 2H+ + 2e™ Ferricyanide + 2e™ NO3 (Nitrate) + 2H+ + 2e Cytochrome ag(+3) + e Fe (+3) + e ¹0₂ + 2H+ + 2e = Reductant Pyruvate + H₂O a-ketoglutarate + H₂O Acetaldehyde + H₂O Ferredoxin (reduced) H₂ Isocitrate B-Hydroxybutyrate Malate NADH NADPH Lipoate (reduced) Glyceraldehyde-3-phosphate + P₁ 2 Glutathione (reduced) FADH₂ Ethanol Lactate Malate O₂ (superoxide) Glutamate + H₂O FADH₂ (enzyme-bound) Methylene blue (reduced) Succinate QH₂ Ascorbate Cytochrome b (+2) Cytochrome c₁ (+2) Cytochrome c (+2) Cytochrome a (+2) H₂O₂ Ferrocyanide NO₂ (Nitrite) + H₂O Cytochrome a3 (+2) Fe(+2) H₂O n 2 2 2 1 2 NN 2 2 2 2 2 2 2 NN 2 2 2 2 2 1 2 2 2 2 2 2 1 1 1 1 2 2 2 1 1 2 E° (V) -0.70 -0.67 -0.60 -0.43 -0.42 -0.38 -0.35 -0.33 -0.32 -0.32 -0.29 -0.29 -0.23 -0.22 -0.20 -0.19 -0.17 -0.16 -0.14 -0 to -0.30 0.01 0.03 0.04 0.06 0.07 0.23 0.25 0.29 0.30 0.36 0.42 0.55 0.77 0.82
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To calculate the equilibrium constant K for the glutathione peroxidase reaction at 37C using the sta...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Biochemistry Concepts And Connections
ISBN: 9780134641621
2nd Edition
Authors: Dean Appling, Spencer Anthony-Cahill, Christopher Mathews
Question Posted:
Students also viewed these Sciences questions
-
Calculate the equilibrium constant for the reaction H2 2H at a temperature of 2000 K, using properties from Table A.8. Compare the result with the value listed in Table A.10.
-
Calculate the equilibrium constant for the reaction H2 2H at a temperature of 2000 K, using properties from Table A.9. Compare the result with the value listed in Table A.11.
-
At what annual interest rate, compounded annually, would $500 have to be invested for it to grow to $1,892.84 in 12 years? Question content area bottom Part 1 The annual interest rate, compounded...
-
Line A: y = 3 0.6x Line B: y = 4 x a. Graph the linear equations and data points. b. Construct tables for x, y, Ëy, e, and e 2 similar to Table 4.4 on page 151. c. Determine which line fits the...
-
Accounting standards are set in the United States in the private sector. Public hearings and written documents provide feedback during development of the standards. Opportunity is provided to those...
-
PepsiCo, inc. (PEP), the parent company of Frito-Lay snack foods and Pepsi beverages, had the following current assets and current liabilities at the end of two recent years: a. Determine the (1)...
-
Photolithography is an important process in integrated circuit manufacture in which a circuit pattern is transferred from a mask onto a photosensitive polymer (the PR), ultimately replicating that...
-
Adam and Arin Adams have collected their personal income and expense information and have asked you to put together an income and expense statement for the year ended December 31, 2012. The following...
-
5. Find the derivative of the following. a. y = 2x+3 x-2 b. y = In (cos(2x-1)) c. y = ex arc sin (2x+3) d. 4x+2xy + y = 0 e. Find the partial derivative with respect to x of the function xy - 5y + 6...
-
Inflammatory stimuli cause macrophages to undergo dramatic metabolic reprogramming, including a switch from oxidative phosphorylation to aerobic glycolysis. Cordes et al. (J. Biol. Chem....
-
Given the roles of NAD + /NADH in dehydrogenation reactions and NADPH/NADP + in reductions, as discussed in Chapter 11 (Section 11.4), would you expect the intracellular ratio of NAD+ to NADH to be...
-
In Exercises 7.27 and 7.29 we compared the effects of two types of feed at a time. A better analysis would first consider all feed types at once: casein, horsebean, linseed, meat meal, soybean, and...
-
1. Does the data for each county indicate the economy is health or unhealthy? How do you know? 2. Describe how the economic indicators affect one another. 3. Which stage of the business cycle is each...
-
Conisder the following three individual protfolions cossisting of investments in four stocks: Stock beta John investment Beth investment jake investment Apple 1.3 2500 5000 10000 Tesla 1.0 2500 5000...
-
The following is Sandhill Photo Tours Limited's unadjusted trial balance at its year end. January 31, 2024. The company adjusts its accounts annually. Cash Accounts receivable Supplies Prepaid rent...
-
Given below are the expected dividends of Kwik Inc. for the next five years. After year five, the dividends are expected to grow at a constant rate of 3% per year forever. Investors require a return...
-
P21.1B (LO 2,4) (Lessee Entries, Finance Lease) The following facts pertain to a non-cancelable lease agreement between Jupiter Leasing Company and Siskiwit Company, a lessee: Commencement date...
-
Accounting practice distinguishes among different types of accounting changes. Identify three different types of accounting changes.
-
Research an article from an online source, such as The Economist, Wall Street Journal, Journal of Economic Perspectives, American Journal of Agricultural Economics, or another academic journal. The...
-
There is a reaction in carbohydrate metabolism in which glucose-6-phosphate reacts with NADP + to give 6- phosphoglucono-d-lactone and NADPH. In this reaction, which substance is oxidized, and which...
-
A biochemical reaction transfers 60 kJ mol -1 (15 kcal mol -1 ) of energy. What general process most likely would be involved in this transfer? What cofactor (or cosubstrate) likely would be used?...
-
The following half reactions play important roles in metabolism. Which of these two is a half reaction of oxidation? Which one is a half reaction of reduction? Write the equation for the overall...
-
A boat travels 30 km north and then 40 km west. a. Make a vector drawing of the path of the boat. b. Draw the vector that represents the net displacement of the boat. c. Determine the magnitude of...
-
% For her initiation into a secret yoga club, Inna is asked to walk on red-hot coals made from firewood. (A) Should she be nervous? Explain. (B) What could Inna do to make this safer (she can't wear...
-
An electrical engineer is attempting to determine the currents through each branch of the circuit in the figure below. The engineer has assumed the currents are in the directions shown in the figure....

Study smarter with the SolutionInn App


