The electrons that reduce nitrate to nitrite are brought to it by a short electron transport chain
Question:
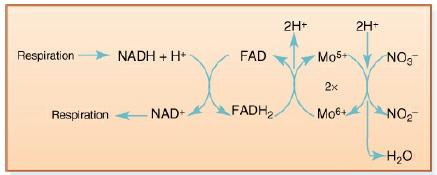
The electrons that reduce nitrate to nitrite are brought to it by a short electron transport chain involving FADH2 and NADH (see Figure 13-16). In which other reactions have you seen these two electron carriers?
Figure 13-16
Transcribed Image Text:
Respiration Respiration NADH + H+ NAD+ FAD FADH₂ 2H+ Mo5+ 2x Mo6+ 2H+ NO₂ NO₂ H₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Citric Acid Cycle Krebs Cycle Both FADH2 and NADH are produced during the citric acid cycle which is ...View the full answer

Answered By

Akash M Rathod
I have been utilized by educators and students alike to provide individualized assistance with everything from grammar and vocabulary to complex problem-solving in various academic subjects. I can provide explanations, examples, and practice exercises tailored to each student's individual needs, helping them to grasp difficult concepts and improve their skills.
My tutoring sessions are interactive and engaging, utilizing a variety of tools and resources to keep learners motivated and focused. Whether a student needs help with homework, test preparation, or simply wants to improve their skills in a particular subject area, I am equipped to provide the support and guidance they need to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Laura Leasing SA signs an agreement on January 1, 2022, to lease equipment to Plote AG. The following information relates to this agreement. 1. The term of the non-cancelable lease is 3 years with no...
-
Pickle Construction Division The Pickle construction division was working on one project for the 2012 fiscal year. They use the percentage of completion revenue recognition method. Contract for new...
-
Consider a memory system with the following parameters: Tc = 100 ns Cc = 0.01 cents > bit Tm = 1,200 ns Cm = 0.001 cents > bit a. What is the cost of 1 MByte of main memory? b. What is the cost of 1...
-
(a) Verify that for both collisions in Figure 8.2 the momentum of the two-cart system remains constant. (b) Verify that both collisions are elastic. Figure 8.2 (a) Soft and (b) hard collisions...
-
In its 2011 annual report, Campbell Soup Company reports beginning-of-the-year total assets of $6,276 million, end-of-the-year total assets of $6,862 million, total sales of $7,719 million, and net...
-
Kazibwe, a businessman does not maintain a complete set of books for his business transactions. The following is a summary of his cash transactions for the year ended December 31, 2015. Receipts Kshs...
-
Name the three steps in the conversion of N 2 into organic nitrogen that is part of a plant. Which type of organism is capable of performing each step?
-
Which essential element is neither a component of rock matrixes nor a contaminant in rock? Although this element is abundant in air, no plant or animal can absorb it from the air and use it. Why not?
-
Explain who are likely to be the winners and losers as a result of talks on carbon dioxide emissions. Use the concepts of game theory to illustrate your argument.
-
What is urban economics and what components of a city usually results in higher demand of a city's real estate. Describe at least three
-
9. What is the force on a 1.25 C charged quarter placed 15 cm from another quarter with -9.86 C of charge? F=_91-9 F- 1910) (1.25x105)(-9:46 X162) (0.15m)2 493N 10. How far away would the quarters in...
-
Identify an area of Financial Services where you feel significant opportunities lie in relation to process management. Why do you feel this?
-
Sam, Mary, and David are expecting to receive cash flow stream of $20,000 for five years, starting in three years. If the relevant discount rate for the entire period is 10%. What is the PV of this...
-
Evaluate the sample results and consider the effect on planned substantive procedures perform a quantitative evaluation to determine whether the failure rate is acceptable for each sample. do not...
-
Gibson Networking develops software for internet applications. The market is very competitive, and Gibson Networkings competitors continue to introduce new products at low prices. Gibson Networking...
-
Dawson Companys balance sheet information at the end of 2019 and 2020 is as follows: Additional information: The company did not issue any common stock during 2020. Required : Next Level Fill in the...
-
Show that (CV/V) T = 0. for an ideal and for a van der Waals gas.
-
In practice, the chlorination of methane often produces many by-products. For example, ethyl chloride is obtained in small quantities. Can you suggest a mechanism for the formation of ethyl chloride?
-
In order to get in shape for mountain climbing, an avid hiker with a mass of 60. kg ascends the stairs in the worlds tallest structure, the 828 m tall Burj Khalifa in Dubai, United Arab Emirates....
-
What is the journal entry to recognize a loss on long-term contracts?
-
Who primarily uses a money order service from a bank?
-
Assume Walter and Jesse agree to form a limited partnership, where Jesse is a general partner and Walter is a limited partner. Pursuant to that partnership, Walter and Jesse purchase a camper in...

Study smarter with the SolutionInn App


