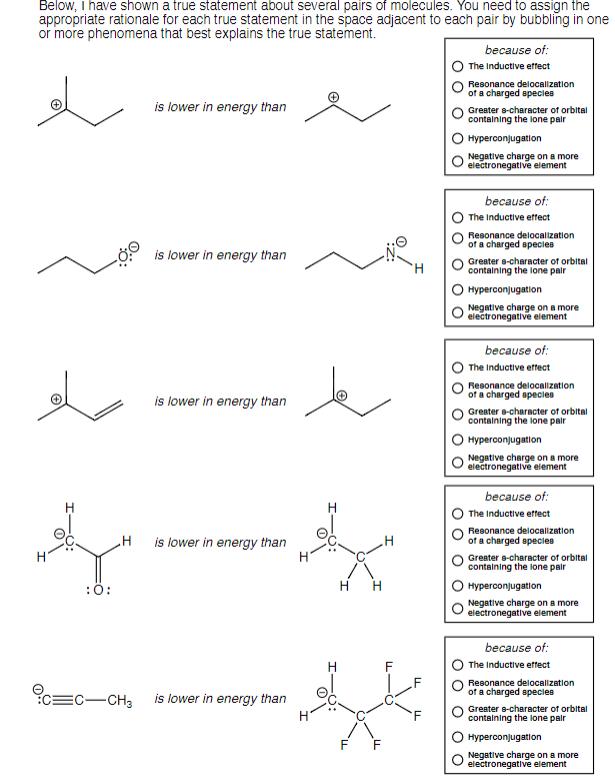
Below, I have shown a true statement about several pairs of molecules. You need to assign...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Below, I have shown a true statement about several pairs of molecules. You need to assign the appropriate rationale for each true statement in the space adjacent to each pair by bubbling in one or more phenomena that best explains the true statement. :O: H EC-CH3 is lower in energy than is lower in energy than is lower in energy than is lower in energy than is lower in energy than H H H H F because of: The Inductive effect Resonance delocalization of a charged species Greater 8-character of orbital containing the lone pair Hyperconjugation Negative charge on a more electronegative element because of: The Inductive effect Resonance delocalization of a charged species Greater a-character of orbital containing the lone pair Hyperconjugation Negative charge on a more electronegative element because of: The Inductive effect Resonance delocalization of a charged species Greater a-character of orbital containing the lone pair Hyperconjugation Negative charge on a more electronegative element because of: The Inductive effect Resonance delocalization of a charged species Greater a-character of orbital containing the lone pair Hyperconjugation Negative charge on a more electronegative element because of: The Inductive effect Resonance delocalization of a charged species Greater e-character of orbital containing the lone pair Hyperconjugation Negative charge on a more electronegative element Below, I have shown a true statement about several pairs of molecules. You need to assign the appropriate rationale for each true statement in the space adjacent to each pair by bubbling in one or more phenomena that best explains the true statement. :O: H EC-CH3 is lower in energy than is lower in energy than is lower in energy than is lower in energy than is lower in energy than H H H H F because of: The Inductive effect Resonance delocalization of a charged species Greater 8-character of orbital containing the lone pair Hyperconjugation Negative charge on a more electronegative element because of: The Inductive effect Resonance delocalization of a charged species Greater a-character of orbital containing the lone pair Hyperconjugation Negative charge on a more electronegative element because of: The Inductive effect Resonance delocalization of a charged species Greater a-character of orbital containing the lone pair Hyperconjugation Negative charge on a more electronegative element because of: The Inductive effect Resonance delocalization of a charged species Greater a-character of orbital containing the lone pair Hyperconjugation Negative charge on a more electronegative element because of: The Inductive effect Resonance delocalization of a charged species Greater e-character of orbital containing the lone pair Hyperconjugation Negative charge on a more electronegative element
Expert Answer:
Answer rating: 100% (QA)
Pair 1 In this pair H2O is the first molecule while H2S is the second Compared to the sulfur atom in H2S the oxygen atom in H2O is more electronegative This implies that the hydrogen atoms will retain ... View the full answer

Related Book For 

Auditing A Practical Approach
ISBN: 9780730382645
4th Edition
Authors: Robyn Moroney, Fiona Campbell, Jane Hamilton
Posted Date:
Students also viewed these mathematics questions
-
Alter's Home Center (AHC) sells renovation and remodeling products to both contractors and individual home owners. One of the services AHC offers is delivery of the purchased products to the...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Jansen, Inc., is a defense contractor that uses job costing. Because the firm uses a perpetual inventory system, the three supporting schedules to the income statement (the schedule of raw materials...
-
On January 1, 2018, Prairie Enterprises purchased a parcel of land for $28,000 cash. At the time of purchase, the company planned to use the land for a warehouse site. In 2020, Prairie Enterprises...
-
Based on the information in Exhibit 1, the most appropriate price-to-earnings ratio to use in the valuation of Delite is closest to: A. 18.71. B. 19.04. C. 24.44. Mark Cannan is updating research...
-
Stonewall Corporation issued \(\$ 20,000\) of \(5 \%, 10\)-year convertible bonds. Each \(\$ 1,000\) bond is convertible to 10 shares of common stock (par \$50) of Stonewall Corporation. The bonds...
-
The stockholders equity accounts of Miley Corporation on January 1, 2014, were as follows. Preferred Stock (7%, $100 par noncumulative, 5,000 shares authorized)....$ 300,000 Common Stock ($4 stated...
-
With the information below, please prepare schedule of cost of goods sold for the three-month period ended March 31, 2019. (Please use this information) Listed below are some of the accounts relating...
-
Can you make the journal entries for the transactions below: Dec. 1 - Sold merchandise to Argem Day Care Center for P20,000 and received a 3-month, 12% note. 2 - Purchased merchandise from Stephen...
-
Q 7 A lessee wants to apply the recognition exemption available for short-term leases. How is the short-term recognition exemption applied? Select the one correct option and then select Submit. By...
-
Does the fact that selling is included as part of the promotional mix weaken or strengthen its role as a sub-element of marketing?
-
Let A and B be events with P (A) = 0.2 and P (B) = 0.9. Assume that A and B are independent. Find P (A and B).
-
Distinguish between a pull and push manufacturing system.
-
Distinguish between value added and non-value added activities.
-
What are committed (locked-in) costs?
-
Chan's new balance is $567.89. He made a payment of $123.45. He made a purchase of $89.00. The finance charge was $2.34. What was the previous balance
-
Which of the ocean zones shown would be home to each of the following organisms: lobster, coral, mussel, porpoise, and dragonfish? For those organisms you identify as living in the pelagic...
-
Canterlot Chartered Accountants is a successful mid-tier accounting firm with a large range of clients across Australia. During 2020 Canterlot gained a new client, Cloudsdale Medical Group (CMG),...
-
Explain why completeness is a more critical assertion for payables than for cash, receivables, inventory, or PPE. What procedures are primarily designed to address the completeness assertion for...
-
Paul Smitten is the partner in charge of the audit for a new client, Umbrella Holdings (UH). The client engaged Pauls audit firm in November 2019, in preparation for the 2020 audit. From 30 January...
-
\(14.5 \pi-5.8 \pi\) Perform the arithmetic operations without a calculator, if possible. If it is not possible, state why.
-
\((7 \sqrt{33}) \times(8 \sqrt{66})\) Perform the arithmetic operations without a calculator, if possible. If it is not possible, state why.
-
\((4 \sqrt{15}) \times(3 \sqrt{10})\) Perform the arithmetic operations without a calculator, if possible. If it is not possible, state why.

Study smarter with the SolutionInn App


