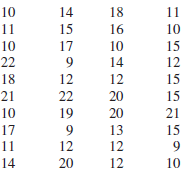
The data show the number of songs on each of 40 CDs from the authors collection. Construct
Question:
Transcribed Image Text:
18 uospS5기59D 으=e202으nuu
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
The distribution is somewhat p...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Elementary Statistics A Step By Step Approach
ISBN: 9781259755330
10th Edition
Authors: Allan G. Bluman
Question Posted:
Students also viewed these Mathematics questions
-
A certain radio station plays songs from the 70's, 80's, and 90's. We know that 20% of the songs on the station are from the 70's; 37% are from the 80's; and 43% of the songs are from the 90's. Let...
-
The English rock band, The Beatles, was formed in Liverpool in 1960. There are 12 studio albums that are considered part of their core catalogue. In the article, "Length of The Beatles' Songs"...
-
The English rock band, The Beatles, was formed in Liverpool in 1960. There are 12 studio albums that are considered part of their core catalogue. In the article, "Length of The Beatles' Songs"...
-
Consider a situation where the marginal damage function is known and equal to D'(E)= d.E. The aggregate marginal abatement cost curve is given by -C'(E)=-bE where is random variable uniformly...
-
McEntire Corporation began operations on January 1, 2016. During its first 3 years of operations, McEntire reported net income and declared dividends as follows. The following information relates to...
-
An equation of motion is given, where is in meters and t in seconds. Find (a) The times at which the acceleration is 0 and (b) The displacement and velocity at these times. 47. s =r-4 - 912 48. s =...
-
What is a system property?
-
LRF Company's budgeted sales and direct materials purchases are as follows. LRF's sales are 40% cash and 60% credit. Credit sales are collected 10% in the month of sale, 50% in the month following...
-
Assume that you have received notice that you are being interviewed for that job. You now have to prepare for the upcoming interview. Part of your preparation is to plan for how you will respond to...
-
Darrow invests $270,000 in cash for a 30 percent ownership interest. The money goes to the original partners. Goodwill is to be recorded. How much goodwill should be recognized, and what is Darrows...
-
Peyton Manning played for the Indianapolis Colts for 14 years. (He did not play in 2011.) The data show the number of touchdowns he scored for the years 19982010. Construct a dotplot for the data and...
-
A local football team has 30 players; the weight of each player is shown. Construct a stem and leaf plot for the data. Use stems 20__, 21__, 22__, etc. 213 202 232 206 219 246 248 239 215 221 223 220...
-
Find each limit. (a) (b) lim x-0 f(x + x, y) - f(x,y)
-
The elementary reaction 2 HO(g) 2 H2(g) + O2(g) proceeds at a certain temperature until the partial pressures of HO, H2, and O2 reach 0.0150 atm, 0.00250 atm, and 0.00650 atm, respectively. What is...
-
At a certain temperature, the Kp for the decomposition of H2S is 0.755. HS(g) H2(g) + S(g) Initially, only H2S is present at a pressure of 0.270 atm in a closed container. What is the total pressure...
-
Carbon disulfide is prepared by heating sulfur and charcoal. The chemical equation is S2(g) + C(s) CS2(g) K = 9.40 at 900 K How many grams of CS2 (g) can be prepared by heating 17.2 mol S2(g) with...
-
Cadmium metal will not reduce zinc ions, but cadmium ions can oxidize manganese metal. Where must the reaction Cd(s) Cd2+ + 2e be located on your table of electrochemical activity?
-
b) The first analyte had a retention time of 7.0 min and a width at the base of 0.30 ml. Further experiments revealed that a non-retained compound had a retention time of 1.9 min. The volumetric flow...
-
The following financial statements were drawn from the records of Culinary Products Co.: Income Statement For the Year Ended December 31, Year 2 Sales revenue...
-
The MIT Sloan School of Management is one of the leading business schools in the U.S. The following table contains the tuition data for the masters program in the Sloan School of Management. a. Use...
-
For the following exercises where applicable: a. State the hypotheses and identify the claim. b. Find the critical value(s). c. Compute the test value. d. Make the decision. e. Summarize the results....
-
A Harris Poll found that 35% of people said that they drink a caffeinated beverage to combat midday drowsiness. A recent survey found that 19 out of 48 randomly selected people stated that they drank...
-
For Exercises perform each of the following steps. a. State the hypotheses and identify the claim. b. Find the critical value(s). c. Compute the test value. d. Make the decision. e. Summarize the...
-
Alice is single and self-employed in 2023. Her net business profit on her Schedule C for the year is $192,800. What are her self-employment tax liability and additional Medicare tax liability for...
-
D.L. Ko and Company, a manufacturer of quality handmade walnut bowls, has had a steady growth in sales for the past 5 years. However, increased competition has led Mr. Ko, the president, to believe...
-
McCullough Hospital uses a job-order costing system to assign costs to its patients. Its direct materials include a variety of items such as pharmaceutical drugs, heart valves, artificial hips, and...

Study smarter with the SolutionInn App


